Introduction
Since the mid-20th century, the livestock and poultry industries have seen advances in several areas including nutrition, genetics, engineering, veterinary medicine, and management, all of which have enhanced feed efficiency, growth performance, and meat yield. Recently, there has been a clamor of calls from industries to focus more on how animal agriculture affects the environment and overall food safety. Similarly, consumers want “clean labels” and to know that their meats are free from residues. They are willing to pay more for "antibiotic-free meats" and will only consume meat from livestock that is raised in a disease free environment (Rummo, 2016).
The global paradigm has shifted from productive efficiency to a far greater focus on public security. This change involves issues concerning the use of antibiotic growth promoters and has occurred because of a focus on greater general safety in our food production systems. For decades, medicated feeds such as antibiotics have been used in animal agriculture to improve growth performance and protect animals from the adverse effects of pathogenic and non-pathogenic enteric microorganisms. Currently, medicines are under increasing scrutiny because of the potential development of antibiotic-resistant human pathogenic bacteria after prolonged use (Phillips, 1999; Ratcliff, 2000). In 1997 the European Union started banning the use of sub-therapeutic levels of antibiotics.
In June of 2003, McDonald’s Corp. announced that after 2004, it would prohibit its poultry suppliers from supplying poultry fed antibiotics (O’Rourke, 2003). Likewise, McDonald’s said it is partnering with beef producers in its top 10 beef-sourcing markets to measure current antibiotic use, and by the end of 2020 will establish reduction targets in those markets. The markets, which represent 85 percent of McDonald’s beef supply chain, will report progress starting in 2022 (Elajalde-Ruiz, 2018). They created a purchasing preference for companies that work to minimize antibiotic use. Meeting market demand for reduced use of antibiotics is crucial for business success and lessens food-animal exposure to antibiotics, which may also reduce antimicrobial resistance over time. Whether or not banning all antibiotic growth promoters is scientifically justified, the tide of public opinion is forcing animal agriculture to develop alternatives, or at least substantially reduce the number of antibiotics used in the production of safe animal products. Some of the other options may include significant changes in husbandry practices and the strategic use of enteric microflora conditioners, including acidifiers, probiotics, enzymes, essential oils, botanicals singly, and in combination.
The Exploding Need for Antibiotic-Free Meat Protein
David Bunge writing in The Wall Street Journal (Bunge, 2015) stated that food producers face a monumental task. At current consumption rates, the world would need to generate 455 million metric tons of meat annually by 2050, when the global population is expected to reach 9.7 billion, from today’s 7.3 billion. Given today’s agricultural productivity, growing the crops to feed all of the poultry, swine, beef and other livestock would require every acre of the planet’s cropland, according to research firm FarmEcon LLC—leaving no room for raising the grain, fruits, and vegetables that humans also need (Bunge, 2015). One approach is to seek ways to breed chickens that will grow faster on less feed and use fewer drugs, according to Dr. Paul Siegel, Distinguished Professor Emeritus at Virginia Tech. Implementation of management strategies and proven technology in animal production systems have provided continual improvement in the production efficiency of animal protein for human consumption. As the use of antibiotics in animal agriculture decreases, there is an increased demand for implementation of technology that continues the increased efficiency in food animal production.
Since the 1950s, antibiotics have been used routinely in the feed and drinking water of livestock raised for human consumption. This practice has resulted in animals being in better health and has encouraged regulatory agencies worldwide to call for a decrease in the use of antibiotics in farm animals due to increasing concerns over antibiotic-resistant superbugs in humans. Antibiotics must be used to treat sick animals; the concern is why they should be used as growth enablers. Sick animals that require antibiotic therapy under the care of a veterinarian should receive the appropriate antibiotics at the prescribed dosage to ensure overall animal health and well-being.
While there is little convincing scientific evidence that the use of antibiotics in food-producing animals is contributing to the resistance issues that are relevant to human medicine, public perception in first-world countries suggests that consumers believe this to be the case. According to the U.S. Organic Trade Association, sales of antibiotic-free organic foods have grown at a rate of 20% per year since 1990. In 2016, The Wall Street Journal reported that poultry producer Pilgrim’s Pride Corporation, the second-largest U.S. poultry producer, announced plans to eliminate all antibiotics from a quarter of its chicken production by 2019 (Bunge, 2015). Five months later, The Journal published that the sale of antibiotic-free beef, while only 5% of the total beef market, has been growing at a pace exceeding the overall market. From 2011–2014, when retail beef sales were up 12.1%, natural beef grew at a rate of 38.9% and organic beef at an astounding rate of 324.1% (Rummo, 2016). This growth in natural and organic beef sales occurred despite a 30%- 80% increase in the costs of raising natural, grass-fed and organic beef (grown without the use of antibiotics) that is ultimately passed along to consumers (Kesmodal, 2015).
Controlling Gut Health in Livestock Without Antibiotics
According to Olsen et al. (2017), it is evident that in today's swine industry, producers must look for alternatives to the use of subtherapeutic growth-promoting antibiotics. The implementation of the Veterinary Feed Directive by the U.S. FDA has shifted the way producers can use antibiotics for growth promotion, which only adds urgency for solutions. Olsen et al. also indicated that the effective use of feed additives to manage gut health is dependent upon their mechanisms of action. The modes of action of growth-promoting antibiotics and the alternatives can differ considerably. Sub-therapeutic antibiotics work in part by decreasing the microbial load in the gut, resulting in a reduction in the amount of energy and protein required to maintain and nourish the intestinal tissues. Croom et al. (2000) reported that because the energy needed to maintain the gut accounts for about 25% of the total basal metabolic needs of an animal, any reduction in gut tissue mass can have a significant impact on the amount of energy available for growth and caloric conversion efficiency. The reduced microbial load in the gut by sub-therapeutic levels of antibiotics also reduces immunological stress (Croom et al., 2000), resulting in more nutrients partitioned toward growth and production rather than toward mechanisms associated with disease resistance.
In contrast, most alternative compounds, such as organic acids, minerals, botanicals and essential oils alone or in combination, do not reduce overall microbial loads in the gut and thus will not promote growth by a mechanism similar to antibiotics. Instead, they alter the gut microflora profile by limiting the colonization of unfavorable bacteria while promoting the fermentation of more favorable species (Dittoe et al., 2018). Consequently, alternatives to antibiotics promote gut health by several possible mechanisms, including altering gut pH; maintaining protective gut mucins; selection for beneficial intestinal organisms; enhancing fermentation acids; enhancing nutrient uptake; increasing the humoral immune responses.
For example, as an antibacterial and antifungal preservative, benzoic acid is widely used in foods and feeds to improve growth and health, which should, at least partially, be derived from the promotion of gut functions, including digestion and absorption. Chen (2015) demonstrated that using appropriate benzoic acid levels might improve gut functions via regulating enzyme activity, redox status, immunity, and microbiota, but that excess administration would lead to the damage of gut health through redox status.
Additionally, the antimicrobial activity of essential oils and their components has been recognized for several years (O’Bryan et al., 2017). Essential oils are produced as secondary metabolites by plants and can be distilled from different portions of plants (Corliss et al. 2017). The recent emergence of bacteria resistant to multiple antibiotics has spurred research for alternatives such as the use of essential oils. These essential oils have beneficial effects for livestock, including reduction of foodborne pathogens (Lee et al. 2008, Lillehoj et al., 2011). Lillehoj (2016) reported that numerous studies had been conducted to study the mode of action of essential oils, and the resulting elucidation of bacterial cell targets has contributed to new perspectives on countering antimicrobial resistance and pathogenicity of these bacteria. Strategic use of these alternative compounds should optimize growth as long as they are used in such a manner that complement their modes of action.
Reducing Enteric Diseases Via Microencapsulation Technology—Studies
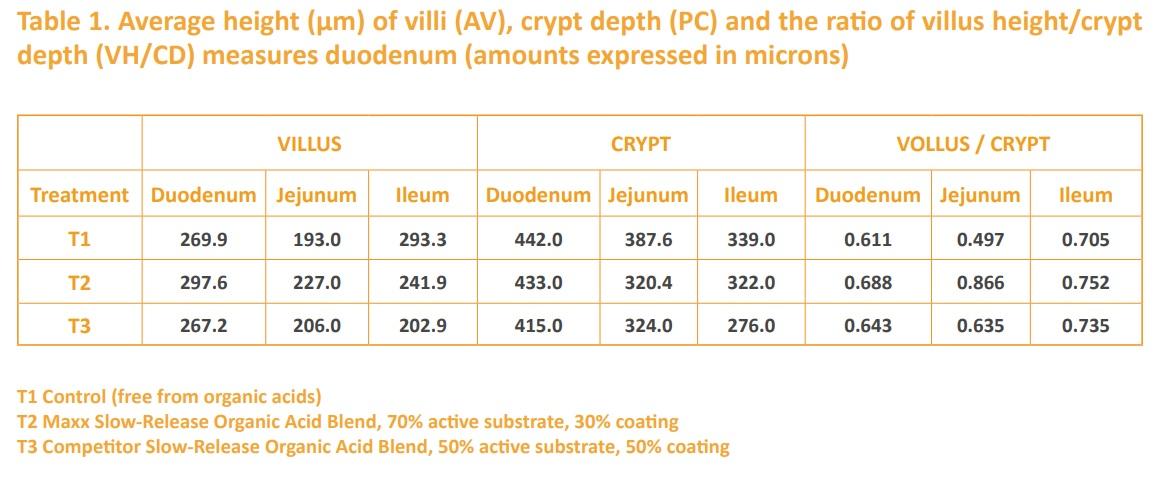
Clostridia and pathogenic coliform bacteria that are often associated with enteric diseases do not grow well in media with a low pH. Therefore, any means to reduce gut pH should improve an animal’s resistance to enteric diseases. Organic acids and their salts are currently presented as an alternative to reduce gastric pH (Miguel et al. 2011). Because organic acids and compounds such as Zinc Oxide have strong bacteriostatic effects, they have been used as salmonella-control agents in feed and water supplies, especially for swine (Linden, 2013). Organic acid blends have also been used as acidifiers in baby pig diets to reduce enteric diseases. The high stomach pH of weaning baby pigs is favorable for the growth of pathogens. At such high pH, gastric digestion is impaired, and there is a risk for the development of pathogenic bacteria and therefore, diarrhea (LI et al., 2008). In a 2014 Brazilian study (Callegari, 2014), a blend of slow-released microencapsulated organic acid, manufactured by Maxx Performance, was shown to increase villi height (Table 1), the production of volatile fatty acids, and the absorptive capacity of intestinal tissues. Additionally, slow-released microencapsulated organic acid blend quantitatively decreased the number of piglets with diarrhea (Table 2).
Recovery time for piglets affected with diarrhea was shorter for groups fed the slow-release organic acid blends versus piglets with diarrhea who were fed the control diets.
In a recent study (unpublished, 2017), Maxx Performance microencapsulated slow-release organic acids, and essential oils were incorporated into the diets of veal calves to investigate growth, disease incidence, and mortality risk. The resulting mortality rate (4/80 deaths in the control group compared 0/80 deaths in the treated group) decreased (P<.05), and the incidence of navel infection was lower in calves fed the slow-release organic acid compared to calves fed the control diet. Wilson et al. (2000) and Pempek et al. (2017) indicated that navel inflammation is a common health concern observed in veal calves when they arrive at the growing facility. Microencapsulated slow-released organic acid and essential oils may be an economical alternative to antibiotic treatment of navel infection in calves. In another study (unpublished, 2018), 300 sows fed microencapsulated slow-release organic acids were compared to 300 sows fed a conventional diet. Sows receiving the slow- release organic acids essential oils diet had a 26% decrease (P<0.01) in piglet mortality from piglet diarrhea compared to sows receiving the regular food. When pigs were supplemented with dietary unprotected Benzoic Acid at the rate of 0.85% and 1%, there was a reduction in the population of E. coli in the intestinal digesta and a decrease in beneficial Lactobacillus organisms (Kluge et al., 2006; Overland et al., 2007). Thus, protecting the Benzoic Acid until its release at the site where E. coli organisms colonize the intestine and should deliver this beneficial organic acid to the gut without affecting the population of beneficial Lactobacillus organisms.
The benefits of using acidic modifiers and organic acids for poultry, however, may be less conclusive. Dietary acidifiers may work better in baby pig diets because they have more limited hydrochloric acid production than chicks. Moreover, dietary organic acids are quickly neutralized in the duodenum unless they are delivered to the ileum and below by adsorbent vehicles. Commercial preparations of slow-release microencapsulated blends of organic acids and slow-release mineral compounds that deliver these non-medicated feed additives to the duodenum and beyond are being used to improve gut health in poultry and thereby lessen the use of sub-therapeutic levels of antibiotics.
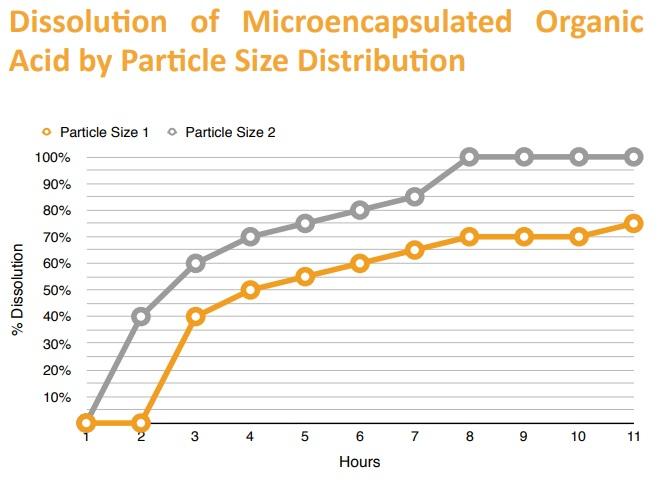
For effective delivery of slow-released compounds, Maxx Performance and collaborators have found that particle size affects the rate of dissolution. For example, as seen in the figure, the goal of a 75-80% targeted release was achieved in 6 hours with particle size 2, which then has more application to animals with simple gastrointestinal tracts (monogastrics), while particle size 1 is more applicable to animals with multiple stomachs (polygastrics).
The critical role of gut microbiota in health, especially gut health, has been gradually recognized. Microencapsulation is a technology that can be used to deliver the optimal food and feed additives, either as a single component or in combination, to improve overall gut health in swine, poultry, dairy animals and humans to help lessen the use of sub-therapeutic medicated additives in animal production.














.jpg&w=3840&q=75)