Conclusions
- The results of this study support the hypothesis that intestinal microbiota compete with the host for metabolism of readily available nutrients such as supplemented amino acids.
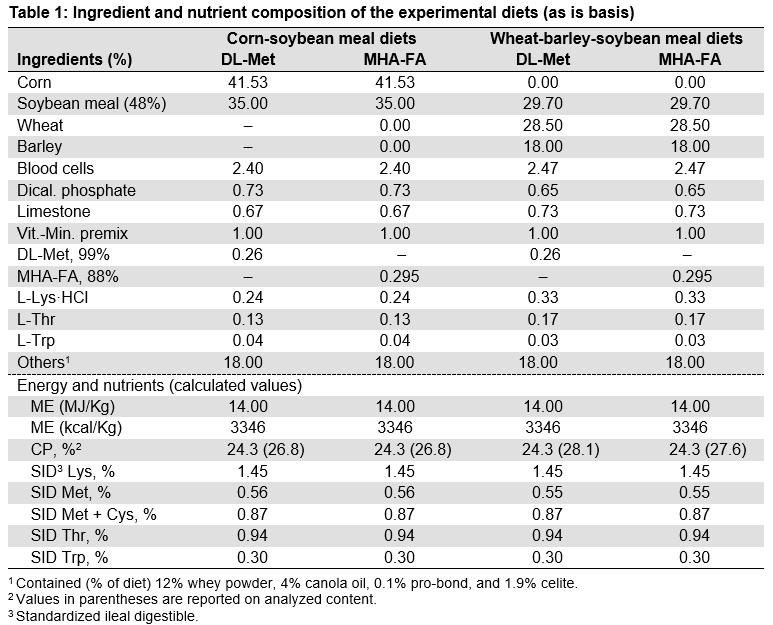
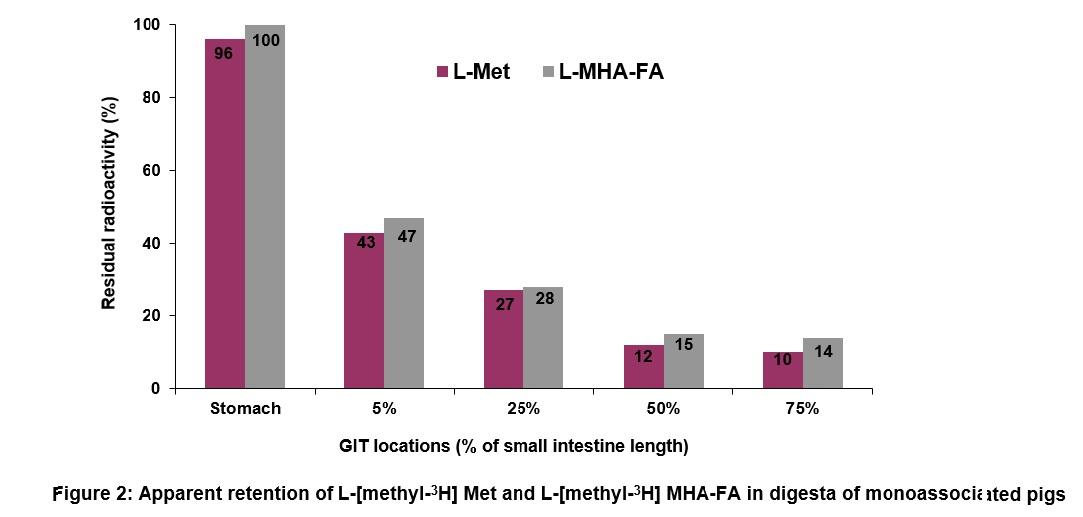
- Apparent residual (unabsorbed) MHA-FA in the small intestine was significantly greater than that of DL-Met in conventional pigs. Furthermore, a higher radioactivity of methionine in the upper small intestine tissue of pigs fed diets supplemented with DL-Met compared with the radioactivity of MHA-FA in pigs fed MHA-FA diets suggests a higher absorption of DL-Met in the small intestine.
- Dietary supplementation of MHA-FA significantly increased total aerobes and lactobacilli populations at 25% of small intestine of conventional pigs, which suggests a preferential degradation metabolism of MHA-FA relative to DL-Met by the gut microbes and is a likely contributor to the lower bioefficacy of MHA-FA relative to DL-Met.
Introduction and objectives
There is increasing realization that microbes in the upper gastrointestinal tract (GIT) compete with the host (pig) for readily available nutrients, including the amino acids (AA; Savage, 1986). It has been reported that various Lactobacillus species, which predominate in pig small intestine (SI), metabolize, i.e. both degrade and produce, AA in in vitro experiments (Liu et al., 2003).
Methionine (Met) is often limiting in swine diets. Supplementation of Met can be accomplished by the addition of DL-methionine (DL-Met; 99%) or liquid form of the DL-Met hydroxy analogue-free acid (MHA-FA; 88%). Drew et al., (2003) showed that intestinal bacteria significantly reduce the absorption of MHA-FA from the GIT of broiler chickens compared to the absorption of DL-Met, and suggested that the lower bioavailability of MHA-FA may be partially attributed by the decreased intestinal absorption of MHA-FA relative to that of DL-Met. It would be interesting and meaningful to determine whether gut microbial modulation also influences the absorption of Met sources in the GIT of pigs.
The main objective of this study was to examine whether microbiota in GIT similarly affect the apparent absorption of Met and MHA-FA in conventional and germ-free (gnotobiotic) pigs. Additionally, the effect of diet formulation based on corn-soybean meal (SBM) or barley-wheat-SBM on the apparent absorption of the two Met sources was also evaluated. This study was conducted by G. Malik and colleagues from the University of Saskatchewan, Saskatoon, Canada.
Experimental design
Dietary treatments included 2 corn-SBM based and 2 wheat-barley-SBM-based diets, supplemented either with DL-Met or MHA-FA on an equimolar basis as Met sources (Table 1). All diets were formulated to contain the same contents of energy and AA which met or exceeded nutrient requirements (NRC, 1998).
A total of 32 conventional and 24 gnotobiotic weaned pigs were used. Conventional pigs were delivered naturally and sow-reared until weaning at 14 days of age. Gnotobiotic pigs were delivered by caesarian section and reared in high efficiency particulate air-filtered isolation units with ad libitum access to a milk-based formula.
Experimental diets were fed to all pigs from 14 to 24 days of age. At 24 days of age, after an overnight fast, pigs were fed at 20 g/kg body weight of experimental diet supplemented with 107 Bq of either L-[methyl-3H] Met or L-[methyl-3H] MHA-FA per kg of diet and chromic oxide (0.5%; wt/wt). Pigs were killed for sample collection 3 hours after consuming the meal, and intestinal contents collected from the stomach and along the length of the SI. Apparent retention of Met and MHA-FA (%) was determined using chromic oxide as indigestible marker according to the following equation:
Apparent retention (%)=(3H activitydigesta: Cr2O3digesta) / (3H activitydiet: Cr2O3diet) ×100
Results
Residual L-[methyl-3H] Met and L-[methyl-3H] MHA-FA activity in contents of conventional and monoassociated pigs are shown in Figures 1 and 2, respectively. In conventional pigs, significantly higher apparent residual (unabsorbed) L-MHA-FA (34% and 15%) compared with L-Met (17% and 8%) were found in digesta recovered at 25% and 75% of SI length, respectively (Figure 1).
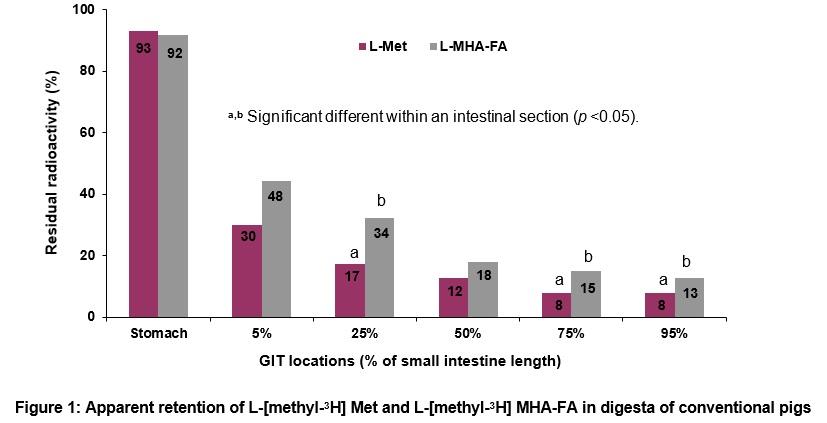
Unfortunately germ-free pigs were contaminated with a single bacterium, i.e. monoassociated; 8 pigs were monoassociated with a gram-negative bacterium closely related to Providencia spp. and 16 pigs were monoassociated with gram positive Enterococcus faecium. Residual L-Met and L-MHA-FA in digesta was not different in monoassociated pigs (Figure 2). Species of monoassociated bacterial contaminant did not affect residual L-Met or L-MHA-FA in the digesta. In both conventional and monoassociated pigs, Met and MHA-FA were retained in stomach (≥ 92%) but disappeared rapidly from proximal SI.
In the upper SI tissue (5% of SI location) of conventional pigs, a significantly higher radioactivity was associated with pigs fed DL-Met-supplemented diets (8.56 ± 0.47 cpm/mg tissue) as compared to MHA-FA diets (5.45 ± 0.50 cpm/mg tissue; Figure 3). These tissue results confirm the greater apparent absorption (lower residual radioactivity) of L-Met from the upper SI (Figure 1). On the other hand, less apparent absorption of MHA-FA from the SI is in line with a lower radioactivity observed in upper SI tissue of pigs fed MHA-FA diets.
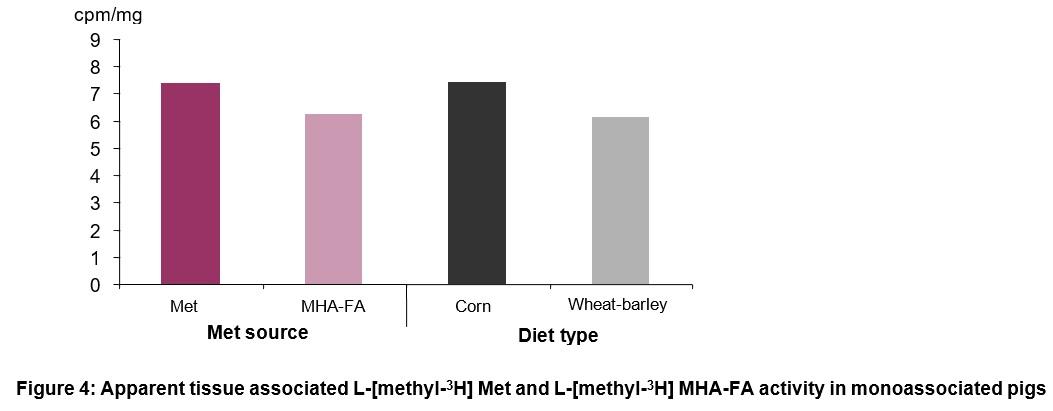
There was no effect of cereal grain type (corn or barley-wheat based diets) on the absorption of methionine sources (Figure 3). The apparent tissue associated activity of L-Met and L-MHA-FA were not different in monoassociated pigs (Figure 4).
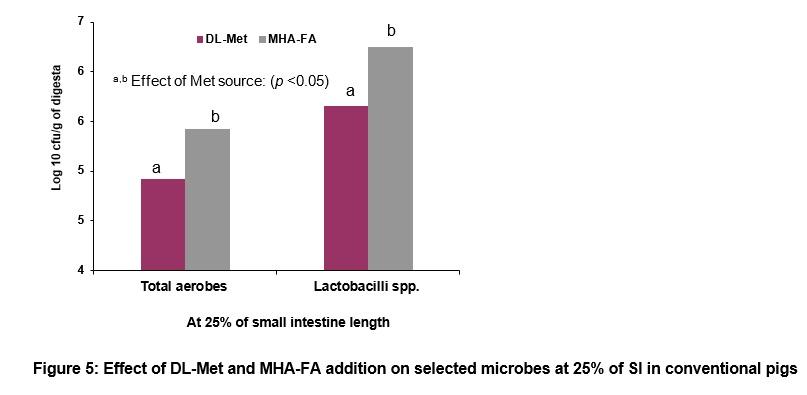
In conventional pigs, supplementation with MHA-FA significantly increased total aerobes and lactobacilli populations at 25% of SI of conventional pigs (Figure 5). This result indicates that gut microbiota can metabolize Met sources differently, and reflects a preferential metabolism (degradation) of MHA-FA relative to DL-Met by the gut microbiota in pigs. This increase in microbial metabolism of MHA-FA associated with a lower amount of MHA-FA available for absorption might contribute to the lower bioavailability of MHA-FA compared with DL-Met.





.jpg&w=3840&q=75)