INTRODUCTION
The Food and Agriculture Organization (FAO) forecasts that the human population will increase by 30% by 2050 (FAO, 2019) with corresponding increase in demand for food. Animal protein, the largest component of human food is entirely dependent on livestock production channels as its source. Over 70% of the cost of livestock production is feed, and the second largest component and cost of feed is the crude protein, a segment that has been challenged for its sufficiency for decades forcing the commercial and scientific communities to be innovative and creative. Several unconventional and less conventional ingredients have been explored as alternative protein sources for livestock in the last several decades.
Hemp (Cannabis sativa L.) is an annual herbaceous plant belonging to the family Cannabinaceae (Turner et al., 1979), traditionally grown for fiber and seed production. Whole hemp seed contains approximately 25% crude protein, 33 to 35% oil, and 34% carbohydrate, in addition to a broad range of vitamins and minerals (Darshan and Rudolph, 2000; Callaway, 2004; House et al., 2010). Hemp seed oil contains 75 to 80% polyunsaturated fatty acids (PUFA), including 60% linoleic acid and 17 to 19% α-linolenic acid (ALA) (Parker et al., 2003). After the extraction of the oil from the seed with a cold press; then the cakes are run through a hammer mill to produce hemp seed cake with a consistent particle size. The nutrient composition of hemp products provides evidence that these products may serve as potentially valuable livestock feed ingredients.
In the past, the cultivation of hemp was prohibited due to the high content of Δ-9 tetrahydrocannabinol (THC) a psychoactive substance present in the hemp plant. In the recent decades, regulatory changes undertaken by several countries across the globe allowed for the legal cultivation of industry hemp under a license that permits plants and plant parts of the genera Cannabis, the leaves and flowering heads of which do not contain more than 0.3% THC, and includes the derivatives of such plants and plant parts. The nutritional profile, in addition to the increase in production and availability of hemp and hemp products create opportunities to use them in livestock diets (Gakhar et al., 2012). Significant research across the globe that has gone into evaluating the safety of the ingredient showed that including hemp in animal feed is safe and offers benefits for improved animal performance and human health (Gakhar et al., 2012; Jing et al., 2017; Kasula et al., 2021c). Initial research indicates hemp products in layers, in addition to the protein contribution, are valuable sources of linoleic acid which is important to improve egg weight (Parker et al., 2003; Silversides and LeFranc, 2005) and linolenic acid and omega fatty acids, which have proven beneficial effects on human health (Lewis et al., 2000; Erasmus, 1993; Silversides et al., 2002; Kasula et al., 2021a). Hemp products are also shown to be excellent sources of yolk pigmentation, lutein and fatty acid enrichment of eggs. Genetic improvements to limit Δ-9 tetrahydrocannabinol to less than 0.3% (w/w) in hemp leaves and flowering heads of the genera Cannabis, have made them safer as a feed ingredient. The use of HSC has not been approved in diets for any class of livestock in the USA due to a lack of research in support of its safety and efficacy. The current study is designed to determine the feeding potential and safety to be used as animal feed ingredient.
Objectives of the study
The objective of the study is to characterize the nutritional and safety properties of HSC as animal feed ingredient as determined by the nutritional (proximate principles, minerals, amino acids and fatty acids) and safety (heavy metal, mycotoxin and cannabinoid) profiles of HSC and its inclusion at increasing levels in finished diets.
MATERIALS AND METHODS
Processing of hemp HSC and location
After the hemp seed is harvested, it is delivered to the processing plant. Upon arrival, it is tested to make sure that it meets quality standards. From there, it is sent through a cleaner to remove foreign material, weed seeds, and other unwanted material. After it is cleaned, it is placed in a storage bin where it is kept until processing. From the storage bin, the hemp seeds move to a cold press to extract the oil. This is a mechanical process so hexane, or other chemicals, are not used in the extraction of the hemp seed oil. The cold press produces a solid cake which is then run through a hammer mill to produce Hemp Seed Cake (HSC) with a consistent particle size. After the product runs through the hammer mill, it is placed in a storage bin for shipment or sent to a bagging facility to be bagged before shipping.
The HSC used in the current study was processed in the lot number 005-2019 and originated from seeds collected from a hemp plant variety CRX-1 which was grown locally, procured and processed by Susquehanna Mills, 349 Village Rd, Pennsdale, PA, USA, with the geographical latitude 40.899938,-77.570296. The feed was manufactured at the Wenger Feed mill located at the 101 West Harrisburg Ave, Rheems, PA 17552.
Study design
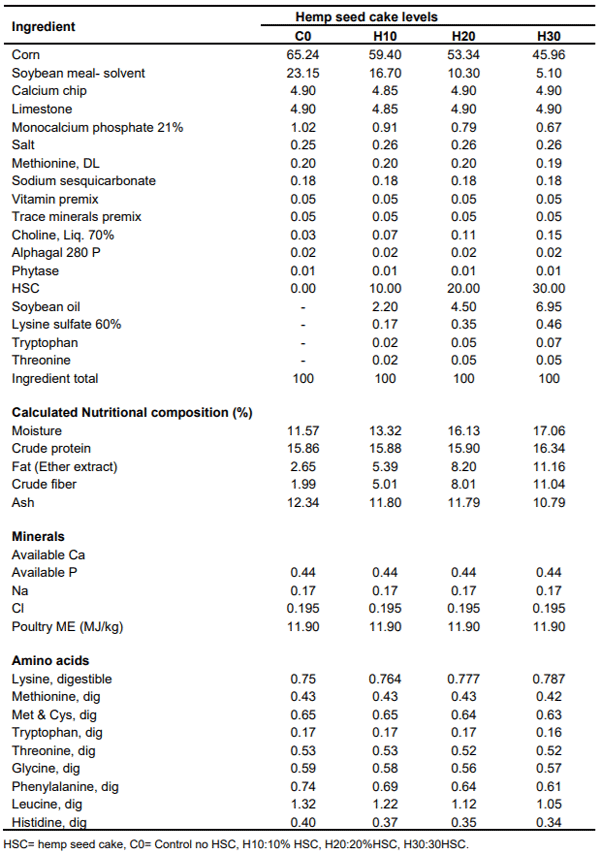
In order to determine the nutritional and safety properties of HSC and its increasing levels in finished diets, the study was designed to analyze the HSC and iso-caloric, and iso-nitrogenous diets containing 16% crude protein, as follows : 1. Control diet – regular diet with no HSC (C0), 2. Regular diet with 10% HSC (H10), 3. Regular diet with 20% HSC (H20) and 4. Regular diet with 30% HSC (H30).
Study parameters
The study parameters were classified under two categories as nutritional and safety. Three samples of every batch of HSC and two samples of each of the four types of finished feed manufactured for the study were analyzed for nutritional parameters comprising of proximate principles including moisture, crude protein, crude fat, crude fiber, total ash, minerals, amino acids and fatty acids analyzed at the New Jersey Feed Lab, Trenton, NJ, and Eurofins Laboratories, (Food Integrity and Innovation-Madison, Madison, WI). The safety parameters included mycotoxins, heavy metals analyzed at the New Jersey Feed Lab, Trenton, NJ, and cannabinoid profile, at Eurofins Laboratories, (Food Integrity and Innovation-Madison, Madison, WI).
Test and analytical methods
Nutritional parameters
Moisture and dry matter (method 925.09 (AOAC, 2000): (i) Set the oven at 80°C before entering the sample
(ii) Weigh 1 g of the ground sample and weigh the paper that will contain the sample.
(iii) Leave samples in the oven for 48 h; weigh the sample at 24 h
(iv) Leave the sample in a desiccator for 10 minutes until the sample reach constant weight,
(v) Use the following formula to calculate moisture and dry matter
(vi) % moisture: ((wet weight-dry weight)/wet weight) × 100
(vii) % dry matter: 100-% moisture
Crude protein (method 990.03 (AOAC, 2000)
(i) Weigh and place 0.5 g of the feed sample in a filter paper
(ii) Transfer the sample in a crystal bottle
(iii) Add 3.5 g of the catalytic mix into the bottle
(iv) Add 8.5 ml of sulfuric acid and then turn on the stove to heat up the bottle with the sample and the catalytic mix for about 90 minutes or until the sample has a green color
(v) Allow the sample to cool down for 5 min;
(vi) Add 25 ml of distilled water and gently move the bottle to spread the water around the bottle (inside).
(vii) Add 30 ml of boric acid and 2 drops of indicator
(viii) Turn on the distiller and open the water to recirculate this
(ix) Transfer sample into the distiller and pour sodium hydroxide into the boiling chamber until it takes a brown coffee color.
(x) Hold the beaker under the distiller and collect not less than 20 ml
(xi) Pour 0.1 N Chloridric acid into the sample until it turns to pink color
(xii) Write down the amount of 0.1 N Chloridric acid spent
(xiii) Calculate the % protein with the following formula:
% protein= spent ml × 0.28× 6.25
Crude fat (method 963.15 (AOAC, 2000)
(i) Weigh 5 g of the sample and weigh the cartridge where the sample will be in
(ii) In a beaker previously weighed add 70 ml of petroleum ether
(iii) Put the beaker in the extractor-digester
(iv) Heat up the extractor until the sample start boiling
(v) Have the sample boing for 6 h and the condensation should be at 60 drops per minute
(vi) After the 6 h, put the beaker to evaporate at room temperature
(vii) After the evaporation, put the beaker in the oven to dry for 1 h and temperature of 80°C
(viii) After 1 hour drying, put the sample in the desiccator and then have a constant weight
(ix) Calculate the % of fat with the following formula:
% fat= (fat net weight/sample weight) × 100
Crude fiber
The crude fiber was determined according to AOAC (2000); briefly two grams of defatted sample were treated successively with boiling solution of H2SO4 of 0.26 N and KOH of 0.23 N. The residue was then separated by filtration, washed and transferred into a crucible then placed into an oven adjusted to 105°C for 18-24 h. The crucible with the sample was weighed and ashed in a muffle furnace at 500°C and weighed. The crude fiber was calculated using the following equation:
CF=W2-W1W3 × 100
Where:
CF= Crude fiber
W1= Weight of crucible with sample before ashing
W2= Weight of crucible with sample after ashing
W3= Weight of sample
Total ash (method 942.05 (AOAC, 2000)
(i) Weigh the container where the sample will be in
(ii) Take 3 g of the ground sample and put into the container
(iii) Put the sample plus the container into the oven at 500°C for 3 h
(iv) After the 3 h, put the sample in the desiccator until the sample reach constant weight
(v) Calculate the % of ash with the following formula:
% ash= (ash weight-weight of the container)/weight of the sample) × 100
Minerals and heavy metals
Mineral Elements by ICP Emission Spectrometry (ICP-OES) (Methods of Analysis of AOAC International, Method 984.27) has been cited by Applegate et al. (2009). The minerals analyzed with the procedure are: Calcium, Ca, Iron, Fe, Sodium, Na, Copper, Cu, Magnesium, Mg, Manganese, Mn, Phosphorous, P, Potassium, K, Zinc, Zn, Aluminum, Al, Barium, Ba, Boron, B, Chromium, Cr, Molybdenum, Mo, Strontium, Sr, Beryllium, Be, Cadmium, Cd, Cobalt, Co, Nickel, Ni, Vanadium, V.
Samples are either dry ashed, wet ashed, or read directly. If dry ashed, the sample is placed in a muffle furnace set to maintain 500°C until ashing is complete. The resulting ash is treated with concentrated hydrochloric acid, dried and re-dissolved in hydrochloric acid solution. If wet ashed, the sample is digested in a microwave or on a hot plate with nitric acid, hydrochloric acid, and/or hydrogen peroxide. The amount of each element is determined with an ICP spectrometer by comparing the emission of the unknown sample against the emission of each element from standard solutions.
The official methods of analysis of AOAC International method 984.27, 985.01, and 2011.14, AOAC International, Gaithersburg, MD, USA (Modified). For the general analysis of component minerals, samples are mineralized by an ashing process at 600°C. The resulting ash is dissolved in mixed acids, diluted as required, and analyzed via ICP-OES (Inductively Coupled Plasma Optical Emission Spectrometry) at (New Jersey Feed Laboratory, Inc., Trenton, NJ).
Amino acids
The samples are hydrolyzed in 6 N Hydrochloric acid for 24 h at approximately 110°C. Phenol is added to the 6N Hydrochloric acid to prevent halogenation of tyrosine. Cystine and Cysteine are converted to S-2-carboxyethylthiocysteine by the addition of dithiodipropionic acid. Tryptophan is hydrolyzed from proteins by heating at approximately 110°C in 4.2 N Sodium Hydroxide. The samples are analyzed by HPLC after pre-injection derivitization. The primary amino acids are derivitized with o-phthalaldehyde (OPA) and the secondary amino acids are derivitized with fluorenylmethyl chloroformate (FMOC) before injection (Schuster, 1988; Henderson et al., 2000).
Fatty acids
The fatty acid composition was determined using standard gas chromatographic techniques of the fatty acid methyl esters (AOAC, 2000, method 969.33), using C17:1 fatty acid (Nu-Chek Prep, Inc., Elysian, MN) as an internal standard. Total lipids were extracted from the HSC and test diets by homogenization in chloroform/methanol (2:1, v/v) according to the methods of Folch et al. (1957). After centrifugation, the organic phase was collected and evaporated under a N2 stream. The all lipid extracts obtained were trans-esterified with methanolysis (1% (v/v) H2SO4 in methanol) for 3 h at 70°C. After cooling, the resulting fatty acid methyl esters (FAMEs) were extracted with hexane and transferred into gas chromatography (GC) vials. All solvents contained 0.005% (v/v) butylated hydroxyanisole (BHA) as an antioxidant. FAMEs were then separated and quantified with a Varian450-GC with CP-8400 autosampler, equipped with a flame ionization detector and a GC column (length 30 m, inner diameter 0.25 mm and film thickness 0.25 μm, DB-225MS) (Agilent Technologies, Mississauga, ON, Canada). Nitrogen was the carrier gas at a column flow rate of 1 ml/min. The inlet split ratio was set at 10:1. The oven temperature programming was as follows: 60°C for 1.5 min, raised to 180°C at 20°C/min, 205°C at 6°C/min, 220°C at 2°C/min for 4 min, and 240°C at 10°C/min for 3 min. The injector and detector temperature were set at 260 and 290°C, respectively. FAMEs were identified by comparison of retention times to known lipid standards (Nu-Chek Prep, Inc., Elysian,Mn) (Folch et al., 1957; Jing et al., 2017).
Safety parameters
Mycotoxins
Mycotoxin concentration was determined by ELISA at the New Jersey Feed Laboratory with the following procedure: The mycotoxin–protein conjugates were adsorbed in separate microplate wells and washed. Samples in solution (50 μl) with a methanol content of 25%, and 50 μl of specific antibodies in PBST (at concentrations of 100, 100, and 500 ng/ml for AFB1, OTA, and ZEA, respectively) were added into the wells and incubated for 8 min with vigorous stirring at room temperature. After washing, a diluted solution of the streptavidin–polyperoxidase conjugate (1:4000 in PBST) was added at 100 μl per well and incubated for 6 min at 37 °C with vigorous stirring. The microplate was then washed four times with PBST, and following 8 min incubation with the substrate solution and the formed immune complexes were detected and quantitatively characterized.
Heavy metals
The levels of heavy metals were determined in HSC and finished feed with the procedure outlined in the mineral procedure section.
Cannabinoids
The levels of cannabinoids in HSC and feed were determined with the following procedure: HSC samples by triplicate and feed samples by duplicate were shipped overnight for the analysis of the residues of various hemp cannabinoids to Eurofins Laboratory, Madison, WI, method 2018.11, by the procedures described in the publication "Quantification of Cannabinoids in Cannabis Dried Plant Materials, Concentrates, and Oils Liquid Chromatography-Diode Array Detection Technique with Optional Mass Spectrometric Detection (Lukas et al., 2018).
Statistical analysis
The safety parameters, mycotoxins, and heavy metals were analyzed with the General Linear Model Procedure (PROC GLM) of SAS (SAS, 2012). The treatment mean separation was carried out with the Tukey Multiple Range test with a probability of error of 5% (P< 0.05). The cannabinoids data were not subjected to statistical analysis because all results below the detectable levels by chromatographic methods in the laboratory.
RESULTS AND DISCUSSION
Nutritional composition of HSC and finished Feed
The analysis of nutritional composition of HSC and feeds formulated with HSC are presented in Table 1. In general, the nutritional composition results were within the expected levels and in agreement with the results in available published literature.
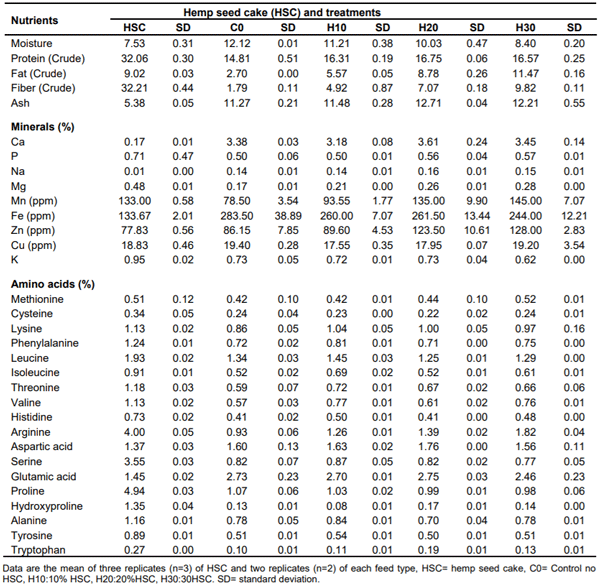
Moisture
The average moisture level of the HSC was 7.53%. This moisture value is in the vicinity of those reported in previous researches, 8.6% in hemp seed meal by Silverside and Lefrancois (2005), 8.8% in HSC (Halle and Schone, 2013) and 9.7% in HSC (Mierliță, 2019). The analyzed moisture levels in the feed were at 12.12% in the control compared to 11.21, 10.03 and 8.40% in the H10, H20 and H30, respectively. This tendency of decreasing moisture level with increasing levels of HSC in finished feed may be attributed to higher dry matter contributed by HSC while replacing corn and soybean meal with soy oil.
Protein
The average analyzed crude protein content of HSC was 32.06%, which is within the range of previous reports, 31.22% reported by Mierliță (2019), and 28.1% by Halle and Schone (2013). The analyzed protein levels of the finished feeds were at 14.81% in the control, 16.31, 16.75 and 16.57% in the H10, H20 and H30, respectively. The analytical variance in crude protein levels in the feed was found to be closer in trends to those reported by Silverside and Lefrancois (2005) who reported crude protein levels of 17.5% in all the control, 5, 10 and 20% HSC treatments in layer feeds when using hemp seed meal, while Halle and Schoene (2013) reported 15.9, 16.5 and 16.9% when HSC was included in the feed at 5, 10 and 15% of the diets, respectively.
Crude fat
The average crude fat levels of HSC were at 9.02%, lower than 12.35% reported by Mierliță (2019) and 11% reported by Halle and Schone (2013). The analyzed levels of crude fat levels in finished feed were at 2.70% in the control, 5.57, 8.78 and 11.47% in the H10, H20 and H30, respectively (Table 1). The higher levels of crude fat with increasing levels of HSC in feed may be attributed to the high level of fat (9.02%) of the HSC compared to the fat content of the major ingredients corn (3.39%) and soybean meal (1.88%) replaced and drawing of soy oil into the formulation as a result of the inclusion of HSC (Table 2).
Table 1. Hemp seed cake and Feed nutritional analysis (% as is basis).
Crude fiber
The crude fiber content of HSC was 32.21%, a value found to be higher than 25.14% reported by Mierliță (2019). The analyzed crude fiber content results of finished feeds were at 1.79 and 4.92% in H10, 7.07% in H20 and 9.82% in H30. This trend of higher crude fiber content with the increasing level of HSC was due to high level of crude fiber (32.21%) of the HSC. These trends of crude fiber agree with those reported by Silverside and Lefrancois (2005) who reported 2.29% in the control feed, 4.22, 6.15 and 10% in layer rations with 5, 10 and 20% hemp seed meal, respectively.
Ash
The ash content of HSC was at 5.38%. Available published literature showed at 7.2% (Silverside and Lefrancois (2005) in hemp seed meal, 6.81% (Mierliță, 2019) and 7.2% (Halle and Schone, 2013) in HSC. The analyzed ash in the feed was at 11.27% in the control, 11.48% in the H10, 12.71% in the H20 and 12.21% in the H30. No published literature was available for comparison of the ash content of the HSC inclusion in feed with different levels in the current study.
Table 2. Study diets formulated by treatment (% in an as is basis).
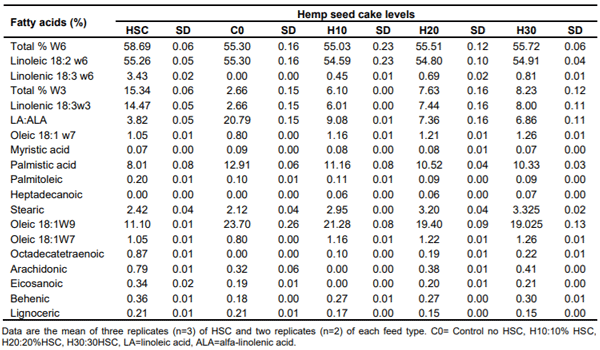
Table 3. Hemp seed cake fatty acid profile (% as is basis).
Minerals
The average analyzed calcium level of HSC was at, 0.17%, found to be lower than 0.28% reported by Halle and Schoene (2013). The analyzed finished feed had a calcium level of 3.38% in the control compared to 3.18, 3.61 and 3.45% when HSC was included at 10, 20 and 30%, respectively (Table 1). The analyzed values of other minerals of HSC and finished feed are presented in Table 1. No published literature was available on these nutrients for comparison purposes.
Amino acids
The average total lysine level in the HSC was at 1.13%, methionine at 0.51% and other amino acids as presented in Table 1. The analysis of finished feeds showed an average total lysine level of 0.86% in control, 1.04% in H10, 1.00% in H20 and 0.97% in H30. Halle and Schone (2013) reported total lysine level of 0.94, 0.87 and 0.85% in layer feed using 5, 10 and 15% HSC, respectively.
The analyzed values of other amino acids of HSC and finished feed are presented in Table 1. No published literature was available on these nutrients for comparison purposes.
Fatty acids
The HSC analyzed for an average total of Omega 6 fatty acids at 58.69% that composed of linoleic acid (18:2 w6) at 55.26% and, linolenic acid (18:3 w6) at 3.43%. The total Omega 3 fatty acid level was 15.34% of which linolenic acid (18:3w3) was 14.47% (Table 3).
In the finished feed, a general increase of omega 3 (total w-3) and individual fatty acids was noticed with increasing levels of HSC; for example, total w-3 values were 2.66, 6.10, 7.63 and 8.23%. The linoleic acid (LA) (18:2 w6) did not present a variance between the HSC treatments; it is at 55.30% in the control feed and 54.595 in the H10, 54.80% in the H20 and 54.91% in the H30. The alfa Linolenic acids (ALA)(18:3 w6 and 18:3 w3) were higher with the increasing levels of HSC; ALAin the control treatment was 2.66% which was increased to 6.01%, 7.44%%, and 8.00% with the H10, H20 and H30, respectively (Table 3). These results make linoleic acid: linolenic acid ratios of 20.79, 9.08, 7.36 and 6.86 in the control and 10, 20 and 30% of HSC. These values are higher than those reported by Mierliță (2019) who reported LA:ALA ratios of 7.98 in the control feed and 3.06 when HSC was included at 20.32% in layer feed. Interestingly, one of the linolenic acids (18:3 w6) was not detected in the control feed and increased with the increasing levels of HSC in the feed from 0 in the control feed to 0.45, 0.69 and 0.81% in the H10, H20 and H30, respectively.
The world population will increase by 30% in the next 25 years (FAO, 2019) and along with that higher population, the demand for food will also be higher. The two most important nutrients in any type of food are protein and energy and both combined represents more than 70% of the food cost; thus, any feed ingredient that can be used to feed animals will contribute to feed the growing human population. The results of this study show that HSC is a rich source of protein (32.06%) and very good source of fat (9.02%) which contributes energy. Additionally, HSC showed to have essential and non-essential amino acids such as lysine and methionine to support egg production and egg weight, respectively. HSC also has a high level of threonine, a very important amino acid to support the immune system; threonine is the highest amino acid found in the mucin amino acid backbone (Gum, 1992), which is a glycoprotein that protects the animal from challenges. L-threonine, cannot be synthesized by humans and animals (Dong et al., 2011; Li et al., 2017; Fang et al., 2020); therefore, any ingredient providing this amino acid is important for animal performance and immunity. Another amino acid found in high level in HSC is proline (4.94%). Although L-proline is not considered to be an essential amino acid in practical poultry diets, chicks fed purified amino acid diets require 0.5% L-proline for optimal growth and feed efficiency (Greene et al., 1962) and it has shown to improve the skin collagen content in poultry (Christensen et al., 1995); other amino acids such as arginine, serine, and alanine are also in high levels in the HSC (Table 1) which are very important for animal feeding and finally to contribute with food security.
Safety of HSC and finished feed
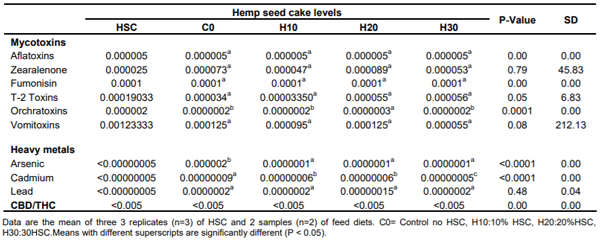
In this study, owing to the large differences in the nutritional values of HSC and experimental diets from published research, the safety of ingredient was addressed by analyzing for heavy metals, mycotoxins and hemp cannabinoid levels. The safety data of the HSC and finished feed represented by mycotoxins, heavy metals are in Table 3; and the cannabinoids in Table 5.
Mycotoxins
The HSC showed an average of aflatoxin 0.000005%, zearalenone 0.000025%, fumonisins 0.0001%, T-2 0.00019033%, ochratoxins 0.000002% and vomitoxins 0.00123333% (Table 4). The levels of mycotoxins were also evaluated in the finished feed with results below the maximum permissible limits. Aflatoxins were below 0.000005 % across all HSC treatments with no significant difference among them. Zearalenone did not show any specific trend with 0.000073% in the control, and 0.000047, 0.000089, and 0.000053% in the H10, H20 and H30, respectively. Fumonisins were recorded at 0.0001% across all treatments. T-2 was 0.000034 % in the control and 0.00003350%, 0.000055 and 0.000056% in the H10, H20 and H30 treatments, respectively (Table 4).
The levels of ochratoxins were recorded to be the same at 0.0000002% in control, H10 and H30 treatments, significantly different from H20 at of 0.00000003% (Table 4). Although the analyzed levels showed a significant difference with H20 treatment, the values were below the permissible limits and did not represent a threat to animals. The average vomitoxin levels did not show significant difference among the treatments and the values ranged from 0.000055 % in the H30 to 0.000125% in control and H20 and 0.000095% in H10 (Table 4).
The maximum level allowed of aflatoxin in an ingredient for poultry is 0.000001% for feed intended for mature animals and not more than 0.000002% if the feed is to be used in immature animals. The maximum permissible levels of Fumonisins are up to 0.001% in an ingredient and not more than 0.0005% in finished feed; Deoxynivalenol (DON) in an ingredient up to 0.0005%b and not more than 0.0002% in finished feed as long as the ingredient is not included at a rate above 50%. No regulatory levels have been specified for the other mycotoxins (FDA, 2020).
Heavy metals
The levels of heavy metals, arsenic, cadmium and lead in HSC and experimental diets are reported in Table 4. The levels of heavy metals in HSC were below laboratory detectable levels of 0.000000005% and were below those reported by several scientists as cited in Table 4. The control ration showed significantly higher levels of arsenic and cadmium over HSC diets. Arsenic was at 0.00000002% in the control, significantly higher than the 0.00000001% observed in all the HSC treatments. Cadmium was recorded at 0.000000009% in the control and was significantly lower at 0.00000000006% in the H10 and H20 and 0.0000000005%in the H30. The lead profiles of experimental rations did not vary significantly.
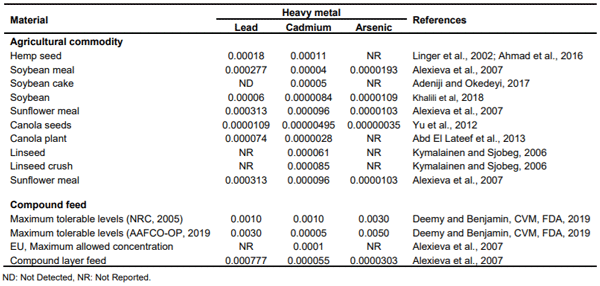
The heavy metal profile of HSC assessed at < 0.000005%for arsenic, lead and cadmium, was lower than published literature on hemp seed or its products and similar conventional processed agricultural commodities such as soybean meal, sunflower meal, canola meal and others (Table 5). Heavy metal levels of various ingredients for animal feeds have been reported to be at 0.000277% lead, 0.00004% cadmium, 0.0000193% arsenic in soybean meal (Alexieva et al 2007); and, 0.000313% lead, 0.000096% cadmium, and 0.0000103% arsenic in sunflower meal (Alexieva et al 2007). Other researchers have found heavy metal values at 0.00018 and 0.0001 mg/kg of lead and cadmium, respectively in hemp seed (Linger et al., 2002). In Linseed and linseed crush, cadmium level was recorded (Kymäläinen and Sjöberg, 2006) at 0.000061 and 0.000085%, respectively.
Table 4. Mycotoxins, heavy metals and CBD/THC in HSC and finished feed (%).
Table 5. Levels of heavy metals in agricultural commodities and compounded feed (%).
Hemp cannabinoids
The hemp cannabinoid levels of HSC and finished diets (Table 4) were reported to be below the detectable levels of 0.005% by chromatographic methods in the laboratory and were under the legal limits of 0.3%. The primary concern with feeding HSC to animals continues to be the transfer potential of hemp cannabinoid residues, mainly cannabidiol (CBD) and delta-9-tetrahydrocannabinol (THC). Published research states that a level of Δ-9 tetrahydrocannabinol (THC), a psychoactive substance in the hemp plant (Health Canada, 2012) below 0.3% is safe for animal feeding (Jing et al., 2017).
Researchers from the European Monitoring Centre of Drug Addiction have reported that HSC may be fed safely to about 30% of the diet to hens (EFSA, 2011). It has been reported that the use of hemp seed up to 30%; up to 10% and up to 20% did not have adverse effects in laying hens (Kasula et al., 2021b).
Most of the published literature on this subject and related areas happens to be with using whole hemp seed or hemp oil or other hemp products. Given the limited published research on the safety of feeding HSC in livestock, the authors are constrained with few supporting references to quote on the findings. The authors have attempted to align with the closest possible references. The current study demonstrates no contribution or transfer of cannabinoids to finished feed from HSC. The safety of any feed ingredient is of upmost importance to prevent potential intoxication, animal health issue, lower performance and the potential food safety issue by passing any residues to humans. The three safety parameters evaluated in this study (mycotoxins, heavy metals, and cannabinoids/THC) were not detected or detected at such lower level that do not represent any threat to livestock or human health. The HSC tested in this trial was later tested in laying hens for a period of 19 weeks, 3 of adaptation and 16 of the experiment, and the levels of these three safety factors were not detected or detected below the legal levels as reported by Kasula et al. (2021a, b, c); these research used the same HSC reported in this study.
Conclusions
The current study has sufficiently evaluated and captured nutritional and safety properties of HSC and finished feeds thereof, with demonstrated conclusions as follows:
(i) HSC presented a nutritional profile consistent with the published literature and contained mycotoxin and heavy metals at levels much lower than permissible.
(ii) HSC did not contain hemp cannabinoids and related compounds detectable by available laboratory methods of analysis.
(iii) HSC could be conveniently accommodated in feed formulations and safely fed up to 30% in commercial laying hen diets.
(iv) HSC may be included as a safe animal feed ingredient.
CONFLICT OF INTERESTS
The authors have not declared any conflict of interest.







_1.jpg&w=3840&q=75)