Aflatoxins: Occurrence, Exposure, and Binding to Lactobacillus Species from the Gut Microbiota of Rural Ugandan Children
Author details:
1 Department of Molecular Cell Biology, Vrije Universiteit Amsterdam, 1081 HZ Amsterdam, The Netherlands; 2 Yoba for Life foundation, 1079 WB Amsterdam, The Netherlands; 3 Department of Medical Biochemistry, School of Biomedical Sciences, College of Health Sciences, Makerere University, P.O. Box 7062, Kampala, Uganda; 4 Department of Nutrition, Institute of Basic Medical Sciences, University of Oslo, 0317 Oslo, Norway; 5 Department of Human Nutrition and Home Economics, Kyambogo University, P.O. Box 1 Kampala, Uganda; 6 Department of Microbiology and Systems Biology, TNO, 3704 HE Zeist, The Netherlands; 7 Institute of Health Sciences, Kristiania University College, 0107 Oslo, Norway; 8 Division of Human Nutrition, Stellenbosch University, Tygerberg, 7505 Cape Town, South Africa; 9 Department of Hematology, Oslo University Hospital, 0318 Oslo, Norway; 10 ARTIS-Micropia, 1018 CZ Amsterdam, The Netherlands.
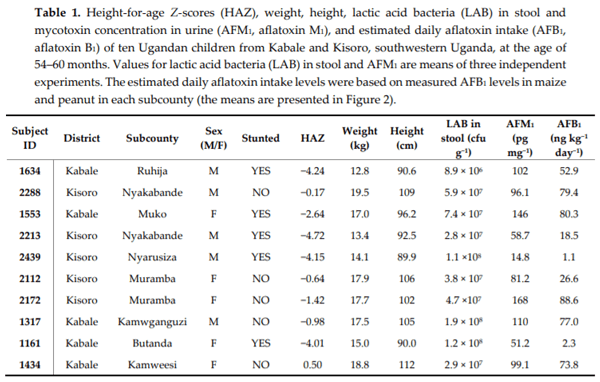
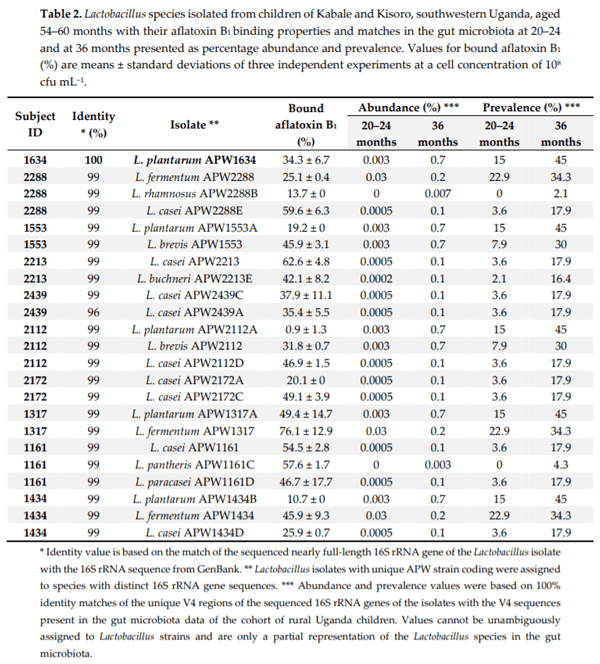
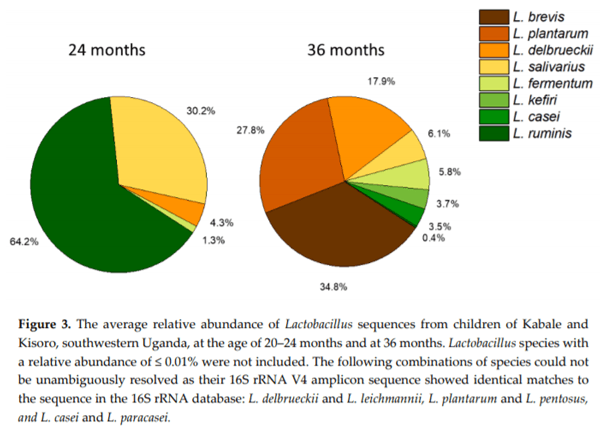
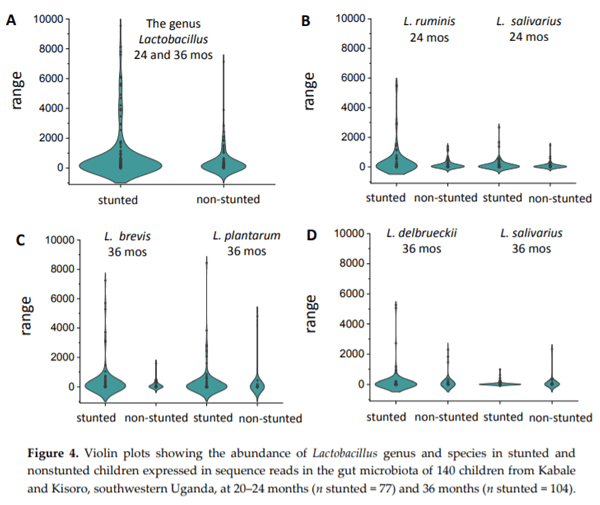
Chronic exposure of children in sub-Saharan Africa to aflatoxins has been associated with low birth weight, stunted growth, immune suppression, and liver function damage. Lactobacillus species have been shown to reduce aflatoxin contamination during the process of food fermentation. Twenty-three Lactobacillus strains were isolated from fecal samples obtained from a cohort of rural Ugandan children at the age of 54 to 60 months, typed by 16S rRNA gene sequencing, and characterized in terms of their ability to bind aflatoxin B1 in vitro. Evidence for chronic exposure of these children to aflatoxin B1 in the study area was obtained by analysis of local foods (maize flour and peanuts), followed by the identification of the breakdown product aflatoxin M1 in their urine samples. Surprisingly, Lactobacillus in the gut microbiota of 140 children from the same cohort at 24 and 36 months showed the highest positive correlation coefficient with stunting among all bacterial genera identified in the stool samples. This correlation was interpreted to be associated with dietary changes from breastfeeding to plant-based solid foods that pose an additional risk for aflatoxin contamination, on one hand, and lead to increased intake of Lactobacillus species on the other.
Keywords: Stunting; aflatoxin B1; Lactic acid bacteria; aflatoxin binding; gut microbiota
1. Chang, W.; Xie, Q.; Zheng, A.; Zhang, S.; Chen, Z.; Wang, J.; Liu, G.; Cai, H. Effects of aflatoxins on growth performance and skeletal muscle of cherry valley meat male ducks. Anim. Nutr. 2016, 2, 186−191.
2. Feng, L.; Yang, D.; Li, H.; Tian, Y.; Zhao, T.; Zhang, R. Survey on pollution of aflatoxin b1 in foodstuffs. Parasitoses Infec. Dis. 2014, 3, 117−119.
3. Wacoo, A.P.; Wendiro, D.; Vuzi, P.C.; Hawumba, J.F. Methods for detection of aflatoxins in agricultural food crops. J. Appl. Chem. 2014, 2014, 1−15.
4. Gong, Y.Y.; Cardwell, K.; Hounsa, A.; Egal, S.; Turner, P.C.; Hall, A.J.; Wild, C.P. Dietary aflatoxin exposure and impaired growth in young children from benin and togo: Cross sectional study. Br. Med. J. 2002, 325, 20–21.
5. Mitchell, N.J.; Hsu, H.-H.; Chandyo, R.K.; Shrestha, B.; Bodhidatta, L.; Tu, Y.-K.; Gong, Y.-Y.; Egner, P.A.; Ulak, M.; Groopman, J.D. Aflatoxin exposure during the first 36 months of life was not associated with impaired growth in nepalese children: An extension of the mal-ed study. PLoS ONE 2017, 12, e0172124, 1−12.
6. Bahey, N.G.; Elaziz, H.O.A.; Gadalla, K.K.E.S. Toxic effect of aflatoxin b1 and the role of recovery on the rat cerebral cortex and hippocampus. Tissue Cell 2015, 47, 559–566.
7. East Africa Standard. Milled Maize (Corn) Products—Specification; East Africa Community: Arusha, Tanzania, 2011; pp. 1–11
8. Codex Alimentarius Commission (CAC) 2014. Codex General Standard for Contaminants and Toxins in Food and Feed. Codex Standard 193–1995. Accessed 14 January 2020 at http://www.codexalimentarius.net.
9. European Communities. Setting maximum levels for certain contaminants in foodstuffs. Commission Regulation (EC) NO. 1881/2006 of 19 December 2006. Off. J. Eur. Union 2006, 364, 5–24.
10. Matacic, C. Fungal toxins are poisoning africa’s children, says new report. Sci. News 2016, 18.
11. Wacoo, A.P.; Wendiro, D.; Nanyonga, S.; Hawumba, J.F.; Sybesma, W.; Kort, R. Feasibility of a novel onsite detection method for aflatoxin in maize flour from markets and selected households in Kampala, Uganda. Toxins 2018, 10, 1−12.
12. Muzoora, S.; Khaitsa, M.L.; Bailey, H.; Vuzi, P. Status on aflatoxin levels in groundnuts in Uganda. Pan Afr. Med. J. 2017, 27, 11.
13. Asiki, G.; Seeley, J.; Srey, C.; Baisley, K.; Lightfoot, T.; Archileo, K.; Agol, D.; Abaasa, A.; Wakeham, K.; Routledge, M.N. A pilot study to evaluate aflatoxin exposure in a rural ugandan population. Trop. Med. Int. Health 2014, 19, 592–599.
14. Kikafunda, J.; Agaba, E.; Bambona, A. Malnutrition amidst plenty: An assessment of factors responsible for persistent high levels of childhood stunting in food secure western Uganda. Afr. J. Food Agric. Nutr. Dev. 2014, 14, 2088–2113.
15. Watson, S.; Gong, Y.Y.; Routledge, M. Interventions targeting child undernutrition in developing countries may be undermined by dietary exposure to aflatoxin. Crit. Rev. Food Sci. Nutr. 2017, 57, 1963−1975.
16. Kumagai, S. Intestinal absorption and excretion of aflatoxin in rats. Toxicol. Appl. Pharmacol. 1989, 97, 88– 97.
17. Liew, W.-P.-P.; Mohd-Redzwan, S. Mycotoxin: Its impact on gut health and microbiota. Front. Cell. Infec. Microbiol. 2018, 8, 60, 1−17.
18. Wang, J.; Tang, L.; Glenn, T.C.; Wang, J.-S. Aflatoxin b1 induced compositional changes in gut microbial communities of male f344 rats. Toxicol. Sci. 2015, 150, 54–63.
19. Galarza-Seeber, R.; Latorre, J.D.; Bielke, L.R.; Kuttappan, V.A.; Wolfenden, A.D.; Hernandez-Velasco, X.; Merino-Guzman, R.; Vicente, J.L.; Donoghue, A.; Cross, D. Leaky gut and mycotoxins: Aflatoxin b1 does not increase gut permeability in broiler chickens. Front. Vet. Sci. 2016, 3, 10, 1–8.
20. Ihekweazu, F.D.; Versalovic, J. Development of the pediatric gut microbiome: Impact on health and disease. Am. J. Med. Sci. 2018, 356, 413–423.
21. Guarner, F.; Malagelada, J.-R. Gut flora in health and disease. The Lancet 2003, 361, 512–519.
22. Li, J.; Sung, C.Y.J.; Lee, N.; Ni, Y.; Pihlajamäki, J.; Panagiotou, G.; El-Nezami, H. Probiotics modulated gut microbiota suppresses hepatocellular carcinoma growth in mice. Proc. Natl. Acad. Sci. U. S. A. 2016, 113, E1306–E1315.
23. El-Nezami, H.S.; Polychronaki, N.N.; Ma, J.; Zhu, H.; Ling, W.; Salminen, E.K.; Juvonen, R.O.; Salminen, S.J.; Poussa, T.; Mykkänen, H.M. Probiotic supplementation reduces a biomarker for increased risk of liver cancer in young men from southern china. Am. J. Clin. Nutr. 2006, 83, 1199−1203.
24. Redzwan, S.M.; Mutalib, M.S.A.; Wang, J.-S.; Ahmad, Z.; Kang, M.-S.; Nasrabadi, E.N.; Jamaluddin, R. Effect of supplementation of fermented milk drink containing probiotic lactobacillus casei shirota on the concentrations of aflatoxin biomarkers among employees of universiti putra malaysia: A randomised, double-blind, cross-over, placebo-controlled study. Br. J. Nutr. 2016, 115, 39–54.
25. El-Nezami, H.; Kankaanpaa, P.; Salminen, S.; Ahokas, J. Ability of dairy strains of lactic acid bacteria to bind a common food carcinogen, aflatoxin b1. Food Chem. Toxicol. 1998, 36, 321–326.
26. Lili, Z.; Junyan, W.; Hongfei, Z.; Baoqing, Z.; Bolin, Z. Detoxification of cancerogenic compounds by lactic acid bacteria strains. Crit. Rev. Food Sci. Nutr. 2018, 58, 2727–2742.
27. Stepman, F. Scaling-up the impact of aflatoxin research in Africa. The role of social sciences. Toxins 2018, 10, 136, 1−16.
28. Wacoo, P.A.; Ocheng, M.; Wendiro, D.; Vuzi, P.C.; Hawumba, F.J. Development and characterization of an electroless plated silver/cysteine sensor platform for the electrochemical determination of aflatoxin B1. J. Sens. 2016, 2015, 1–8.
29. Wacoo, A.P.; Mukisa, I.M.; Meeme, R.; Byakika, S.; Wendiro, D.; Sybesma, W.; Kort, R. Probiotic enrichment and reduction of aflatoxins in a traditional African maize-based fermented food. Nutrients 2019, 11, 265, 1−15.
30. Muhoozi, G.K.; Atukunda, P.; Diep, L.M.; Mwadime, R.; Kaaya, A.N.; Skaare, A.B.; Willumsen, T.; Westerberg, A.C.; Iversen, P.O. Nutrition, hygiene, and stimulation education to improve growth, cognitive, language, and motor development among infants in uganda: A cluster-randomized trial. Matern. Child Nutr. 2018, 14, e12527, 1−11.
31. Atukunda, P.; Muhoozi, G.K.; van den Broek, T.J.; Kort, R.; Diep, L.M.; Kaaya, A.N.; Iversen, P.O.; Westerberg, A.C. Child development, growth and microbiota: Follow-up of a randomized education trial in Uganda. J. Glob. Health 2019, 9, 1−11.
32. WHO Multicentre Growth Reference Study Group. Who child growth standards based on length/height, weight and age. Acta Paediatr. Suppl. 2006, 450, 76, 1−11.
33. Obade, M.I.; Andang’o, P.; Obonyo, C.; Lusweti, F. Exposure of children 4 to 6 months of age to aflatoxin in Kisumu county, Kenya. Afr. J. Food Agric. Nutr. Dev. 2015, 15, 9949–9963.
34. Dary, O.; Jariseta, Z.R. Validation of dietary applications of household consumption and expenditures surveys (hces) against a 24-hour recall method in Uganda. Food Nutr. Bull. 2012, 33, S190–S198.
35. International Conference on Harmonization, Guideline. In Validation of analytical procedures: Text and methodology Q2 (R1). In Proceedings of the International Conference on Harmonization, Geneva, Switzerland, November 2005, pp. 11–12
36. Vasiliades, J. Reaction of alkaline sodium picrate with creatinine: I. Kinetics and mechanism of formation of the mono-creatinine picric acid complex. Clin. Chem. 1976, 22, 1664−1671.
37. Remer, T.; Neubert, A.; Maser-Gluth, C. Anthropometry-based reference values for 24-h urinary creatinine excretion during growth and their use in endocrine and nutritional research. Am. J. Clin. Nutr. 2002, 75, 561–569.
38. Wang, W.; Du, C.; Lin, L.; Chen, W.; Tan, L.; Shen, J.; Pearce, E.N.; Zhang, Y.; Gao, M.; Bian, J. Anthropometry-based 24-h urinary creatinine excretion reference for Chinese children. PLoS ONE 2018, 13.
39. Felske, A.; Wolterink, A.; Van Lis, R.; Akkermans, A.D. Phylogeny of the main bacterial 16S rRNA sequences in Drentsche Aa grassland soils (The Netherlands). Appl. Environ. Microbiol. 1998, 64, 871–879.
40. Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Molecul. Biol. 1990, 215, 403–410.
41. Flach, J.; Ribeiro, C.d.S.; van der Waal, M.B.; van der Waal, R.X.; Claassen, E.; van de Burgwal, L.H. The nagoya protocol on access to genetic resources and benefit sharing: Best practices for users of lactic acid bacteria. Pharma Nutr. 2019, 100158, 1–6.
42. Oksanen, J.; Blanchet, F.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, R.; Simpson, G.; Solymos, P.; Stevens, M.; Wagner, H. Vegan: Community ecology package version 2.3–2. Available online: https://cran.rproject.org/web/packages/vegan/index.html (accessed on 1 September 2019).
43. Siwela, A.H.; Siwela, M.; Matindi, G.; Dube, S.; Nziramasanga, N. Decontamination of aflatoxincontaminated maize by dehulling. J. Sci. Food Agric. 2005, 85, 2535–2538.
44. Okoth, S. Improving the evidence base on aflatoxin contamination and exposure in africa. CTA: 2016.
45. Liu Y, Wu F: Global burden of aflatoxin-induced hepatocellular carcinoma: a risk assessment. Environ. Health Perspect. 2010, 118, 818.
46. Sulaiman, S.H.; Jamaluddin, R.; Sabran, M.R. Association between urinary aflatoxin (AFM1) and dietary intake among adults in hulu langat district, selangor, malaysia. Nutrients 2018, 10, 460.
47. Peltonen, K.; El-Nezami, H.; Haskard, C.; Ahokas, J.; Salminen, S. Aflatoxin b1 binding by dairy strains of lactic acid bacteria and bifidobacteria. J. Dairy Sci. 2001, 84, 2152–2156.
48. Fazeli, M.R.; Hajimohammadali, M.; Moshkani, A.; Samadi, N.; Jamalifar, H.; Khoshayand, M.R.; Vaghari, E.; Pouragahi, S. Aflatoxin b1 binding capacity of autochthonous strains of lactic acid bacteria. J. Food Prot. 2009, 72, 189−192.
49. Lewis, L.; Onsongo, M.; Njapau, H.; Schurz-Rogers, H.; Luber, G.; Kieszak, S.; Nyamongo, J.; Backer, L.; Dahiye, A.M.; Misore, A. Aflatoxin contamination of commercial maize products during an outbreak of acute aflatoxicosis in eastern and central Kenya. Environ. Health Perspect. 2005, 113, 1763−1767.
50. Haskard, C.A.; El-Nezami, H.S.; Kankaanpää, P.E.; Salminen, S.; Ahokas, J.T. Surface binding of aflatoxin b1 by lactic acid bacteria. Appl. Environ. Microbiol. 2001, 67, 3086–3091.
51. Winnie-Pui-Pui Liew, Z.N.; Adilah, L.T.; Sabran, M.-R. The binding efficiency and interaction of lactobacillus casei shirota toward aflatoxin b1. Front. Microbiol. 2018, 9, 1−12.
52. Milani, C.; Duranti, S.; Bottacini, F.; Casey, E.; Turroni, F.; Mahony, J.; Belzer, C.; Palacio, S.D.; Montes, S.A.; Mancabelli, L. The first microbial colonizers of the human gut: Composition, activities, and health implications of the infant gut microbiota. Microbiol. Mol. Biol. Rev. 2017, 81, e00036–e00017.
53. Ismail, M.A.; Taligoola, H.K.; Nakamya, R. Mycobiota associated with baby food products imported into uganda with special reference to aflatoxigenic aspergilli and aflatoxins. Czech Mycol. 2008, 60, 75–89.
54. Kitya, D.; Bbosa, G.; Mulogo, E. Aflatoxin levels in common foods of south western uganda: A risk factor to hepatocellular carcinoma. Eur. J. Cancer Care 2010, 19, 516–521.
55. Liew, W.-P.-P.; Mohd-Redzwan, S.; Than, L.T.L. Gut microbiota profiling of aflatoxin b1-induced rats treated with lactobacillus casei shirota. Toxins 2019, 11, 49, 1−16.
56. Saran, S.; Gopalan, S.; Krishna, T.P. Use of fermented foods to combat stunting and failure to thrive. Nutrition 2002, 18, 393–396.
57. Ahlberg, S.H.; Joutsjoki, V.; Korhonen, H.J. Potential of lactic acid bacteria in aflatoxin risk mitigation. Int. J. Food Microbiol. 2015, 207, 87−102.
58. Lindsay, B.; Oundo, J.; Hossain, M.A.; Antonio, M.; Tamboura, B.; Walker, A.W.; Paulson, J.N.; Parkhill, J.; Omore, R.; Faruque, A.S. Microbiota that affect risk for shigellosis in children in low-income countries. Emerg. Infect. Dis. 2015, 21, 242–250.
59. Wang, R.; Jiang, L.; Zhang, M.; Zhao, L.; Hao, Y.; Guo, H.; Sang, Y.; Zhang, H.; Ren, F. The adhesion of Lactobacillus salivarius REN to a human intestinal epithelial cell line requires s-layer proteins. Sci. Rep. 2017, 7, 44029, 1−10.
60. Peles, F.; Sipos, P.; Gyori, Z.; Pfliegler, W.P.; Giacometti, F.; Serraino, A.; Pagliuca, G.; Gazzotti, T.; Pócsi, I. Adverse effects, transformation and channeling of aflatoxins into food raw materials in livestock. Front. Microbiol. 2019, 10, 2861.
61. Gratz, S.; Mykkänen, H.; Ouwehand, A.; Juvonen, R.; Salminen, S.; El-Nezami, H. Intestinal mucus alters the ability of probiotic bacteria to bind aflatoxin b1 in vitro. Appl. Environ. Microbiol. 2004, 70, 6306–6308
62. Sybesma, W.; Kort, R.; Lee, Y.-K. Locally sourced probiotics, the next opportunity for developing countries? Trends Biotechnol. 2015, 33, 197–200.