Understanding the processes of protein degradation in forage crops provides opportunities for improved silage quality and enhanced animal production
UK silage production has increased from some 0.35 million tonnes in 1947 to 50 million tonnes in 1997. Silage now accounts for almost 80% of the total winter forage fed to ruminant livestock. In Europe, grass accounts for 54%, maize 32%, whole crop cereals 2% and legumes 1% of the crops harvested for silage. Silage making offers many advantages over conventional hay making, such as reduced dependence on weather, increased flexibility of conserving the crop at optimum quality and mechanisation from harvest to feeding.
However, harvesting and storing wetter crops will necessitate more sophisticated machinery and storage facilities than for hay or other concentrate feeds. In particular, greater attention will need to be given to the process of preservation ensuring good compaction, anaerobic conditions and possibly additives to control the fermentation process so as to reduce in-silo losses.
In the UK, prohibition in the use of animal proteins subsequent to the BSE crisis has produced a gap in the supply of protein to ruminants. Concurrently, research evidence has shown that only 5-20% of the nitrogen (N) consumed by ruminants is being recovered in meat or milk. There is therefore the potential for large losses to the environment. Jarvis et al. (1996) calculated that the nitrate-N content in water drainage from a typical dairy farm was above the EU limit for most of the winter. The authors also drew attention to high losses to the atmosphere of ammonia and nitrous oxide, with potentially adverse environmental effects. Approaches to restrict loss of N from the animal have obvious appeal not only to limit environmental pollution but also to improve the economics of animal production.
Figure 1 indicates that more than 70% of the crude protein (CP) consumed by ruminants in Britain is from grassland feeds, with some 14% from cereals, 10% from oilseeds and only a trivial quantity from peas and beans. While the opportunity exists to increase production of CP at low cost this would not necessarily solve the problem of protein supply, because of inefficient utilisation of N by the animal. For example, increasing N fertiliser application to grassland from the current average level of 120 kg N/ha to 300 kg N/ha would double CP production from grassland (calculated from Morrison et al., 1980) through increased yields of dry matter (DM) and increased CP concentration in the DM. This additional CP may not completely replace all of the other feed sources of N, because of limitations to feed intake and inefficient utilisation of CP in grassland feeds.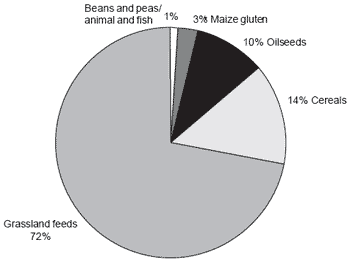
Figure 1. Supply of crude protein to ruminants in Britain in 1995 (thousand tonnes) (from Entec, 1997).
This paper will discuss opportunities for improving the efficiency of utilisation of CP in grassland feeds and the potential for use of high protein alternative forage crops.
Understanding protein degradation in forage and silage The role of nitrogenous compounds in silage has been the subject of a review by Ohshima and McDonald (1978). Of the total nitrogen (TN) containing substances in fresh herbage, some 75-90% are in the form of protein. The remainder consists of soluble nitrogenous organic compounds including free amino acids, amides, glutamine and asparagine together with smaller quantities of amines, chlorophyll, urea products and low molecular weight peptides.
Fresh forage also contains variable amounts of inorganic soluble non-protein nitrogen (NPN) present mainly as nitrate and to a much lesser extent ammonia. The concentration of the NPN products will depend on timing and extent of applied nitrogen-based fertilizers. Concentration of nitrate in fresh forage can be as high as 10% of the total N (McDonald et al., 1991), while ammonia-N concentrations in fresh herbage are usually less than 1.5% (Brady, 1960). Keady (1998) concluded that while increasing N fertiliser application on grassland increased yield of DM and consequently quantity of CP, this was accompanied by a reduction in feed value as intake and performance were depressed when conserved as silage as shown in Table 1.
Table 1. Effects of fertiliser N on animal performance.*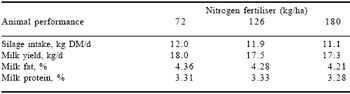
*Adapted from Keady (1998).
PROTEIN DEGRADATION IN SILAGE
Proteolysis during ensilage has been widely reported. Protein content can decrease from 800 g/kg to 300 g/kg during ensilage (McDonald et al., 1991). Protein breakdown commences after mowing and during wilting of the plant material usually as a consequence of plant enzyme activity. Messman et al. (1994) studied the effect of wilting on buffer-extractable proteins from a range of species using S-dodecyl sulphate polyacrilamide gel electrophoresis (SDS-PAGE). No loss of proteins was observed following a 24 hr wilt while a reduction of 30% was detected after five days drying.
The degradation of protein was also accompanied by an increase in NPN content of the extended wilted material. Other workers (Mangan et al., 1991; Carpintero et al., 1979) observed minimal loss of RUBISCO, the major plant leaf protein fraction in wilted silage.
Studies with gammairradiated plant material suggest that hydrolysis (i.e. breakdown to smaller peptides and free amino acids) is largely mediated by plant enzyme proteolytic activity (Oshima and McDonald, 1978). However some strains of bacteria, Lactobacillus plantarum and L. paracasei subspecies paracasei, have also been demonstrated to possess some proteolytic activity (Hickey et al., 1983; Sasaki et al., 1995). Both plant and microbial activities contribute to further catabolism of amino acids (Oshima and McDonald, 1978). Increased concentrations of alanine are frequently observed in small scale studies; and it is postulated that this is due to decarboxylation of aspartic acid (Kemble, 1956; Oshima et al., 1979; Heron et al., 1986). Increases in alanine have also been observed in farm-scale ryegrass silages made at the Institute of Grassland and Environmental Research (unpublished data).
Protein degradation during ensilage consistently results in decreased concentrations of glutamic acid, arginine and aspartic acid and increased concentrations of gamma amino butyric acid and ornithine (Lessard et al., 1978; Oshima et al., 1979; Heron et al., 1986). A simplified illustration of the protein breakdown products of herbage and ensiled material is shown in Figure 2.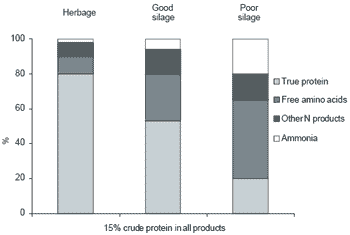
Figure 2. Protein fractions of herbage and silage.
It is clear that the most important protein fraction for efficient ruminant utilisation is the true protein (TP) component. It has also been shown (Jones, 1998) that poorly fermented silages will be associated with higher levels of free amino acids and reduced true protein. It is becoming apparent that routine silage analysis of crude protein does not provide adequate sensitivity for realistic feed formulation purposes. As shown in Figure 2, a similar crude protein value was obtained for the fresh grass herbage as for the poorly and well fermented silage. The free amino acids however, gave a better indication of the protein degradation. Unfortunately the chemical analysis procedures for the estimation of free amino acids is both laborious and expensive. Recently a new method was described by Winters et al. (1999a) where free amino acids from a crude extract of silage using a ninhydrin colorimetric technique was compared with conventional amino acid analysis. The new rapid technique correlated well with conventional amino acid analysis (r2 = 0.87), and provides scope for further development as a routine method of predicting true protein content of silages.
IMPROVING EFFICIENCY OF UTILISATION FROM GRASSLAND FEEDS
The concentrations of CP in grazed grass, silage and grass hay are normally in the range 150-220, 100-160 and 80-120 g/kg DM, respectively, varying seasonally with N fertiliser rate, stage of maturity and the magnitude of losses during conservation. High CP levels in grassland feeds can lead to rapid breakdown in the rumen, leading to high concentrations of rumen ammonia and low levels of undegraded dietary protein (UDP). Low supply of energy to the rumen can also limit microbial protein synthesis, which can restrict the supply of essential amino acids to grass-fed animals. These effects may be even greater in silages as true protein is metabolised to free amino acids, peptides and ammonia as a consequence of proteolysis during the fermentation process.
Beever et al. (2000) noted that the yield of microbial protein with silages varied from 13 to 28 g microbial N/kg organic matter apparently digested in the rumen for silages compared with values of 33-58g for fresh forages. Microbial protein could be increased by improving the supply of readily available energy in feed and/or by improving the synchrony in the supply of N and energy to rumen microbes. In temperate grasses the major source of readily available energy is from water soluble carbohydrates (WSC) with concentrations in fresh grasses varying from 50-350 g/kg DM. There are characteristic differences in WSC seasonally (low in spring and autumn), with stage of growth (high during stem development) and among species (higher in ryegrass than in other sown species).
Humphreys (1989) demonstrated that WSC content is heritable and varieties of perennial ryegrass have been bred with markedly higher WSC content. In a recent trial conducted at IGER where high sugar grasses were fed as zero-grazed to dairy cows, Miller et al. (1999) reported that milk yields were 3 kg/day higher for cows fed the experimental high sugar ryegrass (200 g WSC/kg DM) compared to the commercially available ryegrass (130 g WSC/kg DM). Differences between the varieties in dry matter intake and digestibility were also observed as shown in Table 2.
Table 2. Milk production from grazed high sugar grasses.*
*Miller et al., 1999.
OPPORTUNITIES FOR LIMITING PROTEOLYSIS IN GRASS SILAGE
Preserving grass as silage allows the possibility of changing the nature of the CP through the use of additives and crop management. Bacterial inoculants have been demonstrated to produce silages with higher proportions of true protein than silages made without additive (Jones, 1998; Merry et al., 2000). It is likely that the more rapid acidification in the early phase of ensiling as a result of the dominance of homofermentative bacteria can inhibit protease activity and thereby reduce protein degradation. Jone (1998) has demonstrated improvements in animal performance with silages made with applied bacterial inoculants.
This improvement in animal performance may be associated with improved efficiency of microbial protein. Sharp et al. (1994) reported an improvement of 33% in the efficiency of microbial protein synthesis when silages made with a biological inoculant were compared with well preserved untreated silage. Silage additives containing formaldehyde or tannins can also influence reductions in protein breakdown as discussed by Beever (1980). However, responses were dependent on the application rate used; and a risk of higher concentrations of indigestible protein led to reduced in vivo digestibility and enhanced faecal N loss.
While grass silage exhibits a high concentration of UDP, supplementation with high protein concentrates has shown positive benefits in milk production responses (Aston et al., 1998). Such an approach will undoubtedly lead to increased losses of N to the environment (Table 3).
Table 3. Effect of increasing crude protein proportion in concentrate feed with ad libitum grass silage.*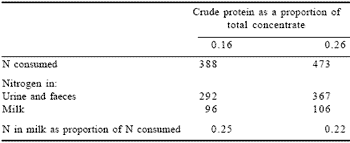
*From Aston et al., 1998.
Role of alternative forage crops
In the post-BSE era alternative forage crops such as legumes, pulses, kale and lupin are assuming a much higher profile in UK agriculture as a consequence of consumer pressure to feed animals on natural, home grown forages. All of these forages are capable of giving relatively high yields of DM and of CP as indicated in Table 4. These forages are also characterised by high levels of feed intake (Beever et al., 2000; Fraser et al., 1999a; b).
Table 4. Protein production from forage crops in the UK.*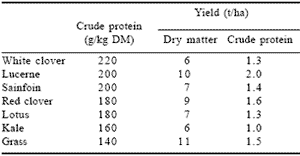
*Adapted from Entec, 1997.
A recent trial conducted by Dewhurst et al. (2000) showed higher intakes and milk yields for silages made from white clover, red clover and lucerne than from grass silage. In addition, levels of intake and production were intermediate when cows were fed mixtures of legume and grass silages as shown in Table 5.
Table 5. Effect of legume silages on feed intake and milk production with cows fed 8 kg/day of concentrates.*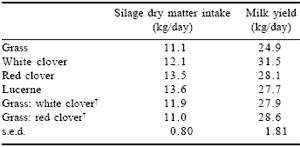
*From Dewhurst et al., 2000.
†Cows fed 1:1 mixtures of silages on DM basis.
It must be noted that while white clover silage performed well in terms of milk production, it is unlikely that a monoculture crop would be economically viable due its low yield (2 tonnes DM/ha). Moorby et al. (1998) found higher levels of performance and milk production from cows fed on kale-barley silage than with grass silage alone.
In relation to protein composition and utilisation there are major differences between these forages. White clover and lucerne true protein (TP:CP) ratios (0.35-0.40) are similar to grass silage with low concentrations of WSC and extensive proteolysis resulting in high concentrations of free amino acids. However, red clover silage has a TP:CP ratio of approximately 0.6. In a comparison of lucerne and red clover silage illustrated in Figure 3 (Winters et al., 1999b), the free amino acids in red clover were markedly lower than for lucerne. Interestingly application of bacterial inoculant to these legume silages gave similar results to previous work conducted with grass silages where further reductions in free amino acids were found.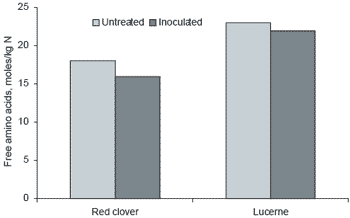
Figure 3. Free amino acids (moles/kg N) in silages made from lucerne and red clover either untreated or treated with a biological inoculant (adapted from Winters et al., 1999b).
While the above studies would indicate that using bacterial inoculant additives provides a level of protein protection and thus higher supply of UDP, Davies et al. (1999) using in vitro rumen simulation technology, concluded that efficiency of microbial N synthesis was 34% higher in untreated red clover silage compared to untreated lucerne silage.
Furthermore, when the red clover silage was treated with a biological inoculant, a further 13% improvement in microbial protein was observed.
Beever and Thorp (1996) concluded that microbial protein yields were higher for silages made from white clover and lucerne than for grass silages, but this arose largely from high CP contents and high levels of feed intake.
High levels of true protein in red clover silages were first noted by Albrecht and Muck (1991) and Jones (1993). The protection of the protein during the fermentation process in the silo is attributed to the high content of polyphenol oxidase (PPO) in red clover. The underlying principles of protein protection have been associated with a reaction of the PPO-generating caffeoquinones with specific proteases (Hatfield and Muck, 1999).
Lotus and sainfoin contain condensed polyphenols (tannins), which may restrict protein breakdown in the silo and in the rumen. Waghorn and Shelton (1997) have provided clear evidence that tannins in Lotus corniculatus grown in New Zealand improve protein utilisation, with reductions in rumen ammonia concentrations by 27%. Associated was an increase in absorption of essential amino acids from the small intestine of 50%, mainly through increased UDP, for which there is a particular requirement with grassbased diets fed to high yielding animals.
In a recent trial conducted in New Zealand (Woodward et al., 1999) with dairy cows in late lactation grazing Lotus corniculatus, significant increases in dry matter intake (14%) and milk production (40%) were observed compared to cows on grazed grass. In order to identify the factors associated with the improvements obtained in the grazing trial, another indoor trial was conducted under a zero-grazing regime where polyethylene glycol (PEG) was dosed daily to half of the cows. The effect of the PEG was to block the proteins from binding to the condensed tannins and effectively forming a by-pass protein. The resulting 43% increase in milk yield by cows fed lotus with no PEG supplement compared to the lotus with PEG supplement indicates that the major effect on improved milk yield was due to the condensed tannin in the lotus rather than total CP supply (Table 6).
Table 6. Milk yield and composition from Friesian cows fed ryegrass or Lotus corniculatus.*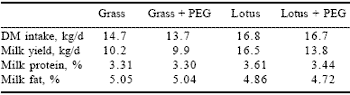
*Adapted from Woodward et al., 1999.
PEG dosed 3 times per day (total 3.6 l/d).
Thomson et al. (1971) demonstrated improved utilisation of protein in dehydrated sainfoin compared to that in dehydrated lucerne and associated this with the condensed tannins in the sainfoin. However, Fraser et al. (1999b) demonstrated that N utilisation of sainfoin silage when fed to growing lambs was low with a negative N balance and more than 80% of the ingested nitrogen was released in faeces. This observation would indicate that a degree of ‘over protection’ of protein was evident and that energy supplementation would not improve utilisation of the sainfoin silage feed.
Red clover offers more opportunities as a high protein crop on UK farms due to its improved agronomic characteristics compared to lotus or sainfoin.
The crop is widely adapted to the soils and climates of northern and western Europe; and in a recent experiment DM yields were higher for red clover than grass with 200 kg N fertiliser/ha at seven out of nine sites in UK, Germany, Sweden and Finland (Halling et al., 2000). The species has limited persistence, but is particularly suited to mixed and organic farming systems. Although of high moisture content and low WSC content, recent research has demonstrated successful ensilage through combination of wilting and use of either biological or chemical additives (Winters et al, 1999b; Pahlow et al., 2000). The CP concentration from forage legumes will have major significance in the diet of animals where predominantly high energy crops such as maize form the basal diet. Increasing efficiency of microbial protein synthesis and UDP in grassland based forages are particularly important in order to maintain competitiveness of the enterprise.
Alternative grain crops
The use of home-produced rapeseed meal is a well-established practice in UK agriculture. The most interesting protein concentrate crops are probably beans, peas and lupins. Beans and peas are traditional crops for much of Britain, with important progress having been made by plant breeders in improving seed yields and harvest index by producing determinant types and, in peas, leafless and semi-leafless varieties. The protein in peas and beans is however readily degraded in the rumen with low levels of UDP (Agricultural and Food Research Council, 1983).
It is unlikely therefore, that these crops would contribute to improved utilisation of protein when used in a mixed diet with grass silage, but could make a major contribution to maize silage-based diets. Lupins are not traditionally grown as a field crop in Britain; and most of the research work carried out to improve varieties has concentrated on autumn sown varieties.
However, maintaining persistency of the lupin crop over winter has been variable. New spring sown varieties of lupins offer greater flexibility of being harvested as an early season crop or allowed to mature as a grain crop. Jones et al. (1999) showed benefits in wilting and application of biological inoculant on the fermentation characteristics of an autumn variety of lupin cv Amiga as shown in Table 7. The relatively high WSC content and low buffering capacity of a lupin forage crop at growth stage 4.7 provided sufficient substrate supply to ensure good preservation.
Application of a bacterial inoculant to lupin silage gave significant benefits in reducing pH from 4.42 to 3.96 and 4.45 to 3.87 and ammonia N from 177 to 130.5 and 131 to 73 g/kg N, in both unwilted and wilted crops either untreated or treated, respectively. The grain in lupin silage has a CP content of ~440 g/kg DM; and Moss and Grundy (1996) found some 0.4 of this to be UDP, approaching the value for soybean meal. The content of antinutritional metabolites is low. There is a need for more direct information on the nutritive value of lupins grown in the UK; but interim results indicate possibilities for substantial replacement of fishmeal and soybean meal in the diets of high yielding cows (Mansbridge and Blake, 1998). With prospects for further improvement through breeding and better agronomy, there is potential for this crop to make a major contribution to ruminant feeding in Britain.
Table 7. Mean composition of lupin silage after 90 days ensiling (all values g/kg DM unless otherwise stated).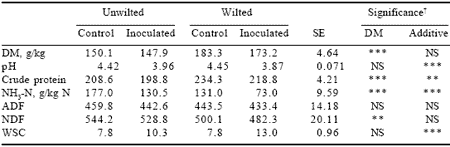
†NS, non-significant, **P<0.01, ***P<0.001.
Conclusions
The supply of CP to ruminants in the UK is dominated by grassland feeds, for which protein is often used inefficiently resulting in large losses to the environment. There are opportunities for improving protein supply from grazed grass herbage by either increasing crop WSC content or reducing rate of protein degradation in the rumen.
The new varieties of high sugar grasses are now available commercially in the UK. In conserved grassland crops the use of biological inoculants to enhance acidification resulting in lower protein degradation offers a realistic and economic return to maximise supply of UDP in ruminants.
Red clover, which is well adapted to UK conditions and has attributes that reduce protein degradation both in the silo and the rumen, has potential to complement grass swards. For grass silage-based rations, the pulse crops, beans and peas, have limitations because of low contents of UDP, but there is high potential for lupins as a seed crop with protein quality and quantity similar in characteristics to soybeans.
Approaches to improve the utilisation of CP in forages and obtaining a good balance between the supplies of N and readily available energy in the rumen are particularly important to reduce input feed costs and minimise losses to the environment.
Acknowledgements
Data supplied from the IGER trials in this paper were commissioned by the Ministry of Agriculture, Fisheries and Food, the Milk Development Council and the European Union. The Institute of Grassland and Environment Research is funded through the Biotechnology and Biological Sciences Research Council. I am indebted to Prof. R.J.Wilkins for his contribution in sourcing some of the more recent research data presented in this paper.
References
Agricultural and Food Research Council. 1983. Energy and Protein Requirements of Ruminants. Wallingford: CAB International.
Albrecht, K.A. and R.E. Muck. 1991. Crop quality and utilisation: proteolysis in ensiled forage legumes that vary in tannin concentration. Crop Sci. 31:464-469.
Aston, K., W.J. Fisher, A.B. McAllan, M.S. Dhanoa and R.J. Dewhurst. 1998. Supplementation of grass silage-based diets with small quantities of concentrates: strategies for allocating concentrate crude protein. Anim. Sci. 67:17-26.
Beever, D.E. 1980. The utilisation of protein in conserved forage. Occ. Symp. 11, Br. Grassld Soc., pp. 131-143.
Beever, D.E. and C. Thorp. 1996. Advances in the understanding of factors influencing the nutritive value of legumes. Occ. Symp. 30, Br. Grassld Soc., pp. 194-207.
Beever, D.E., N. Offer and E.M. Gill. 2000. Feeding value of grass and grass products. In: Grass, Its Production and Utilization (A. Hopkins, ed.), Blackwell Science, Oxford, pp. 140-195.
Brady, C.J. 1960. Redistribution of nitrogen in grass and leguminous fodder plants during wilting and ensilage. J. Sci. Food Agric. 11:276-284.
Carpintero, C.M., A.R Henderson and P. McDonald. 1979. Effect of wilting on ensiling characteristics. Grass Forage Sci. 34:311-315.
Davies, D.R., A.L. Winters, D.K. Leemans, M.S. Dhanoa and R.J. Merry. 1999. The effect of inoculant treatment of alternative crop forages on silage quality and in vitro rumen function. In: Proceedings of the X11 International Silage Conference (T. Pauly, ed), Uppsala, Sweden, pp. 131- 132.
Dewhurst, R.J., D.W.R. Davies, W.J. Fisher, J. Bertilsson and R.J. Wilkins. 2000. Intake and milk production responses to legume silages offered to Holstein-Friesian cows. Proc. Br. Soc. Anim. Sci. (in press).
Entec, 1997. Home grown protein sources for animal feeds. Technical Report to Ministry of Agriculture, Fisheries and Food, Scottish Office Agriculture, Environment and Fisheries Department, Milk Development Council, Meat and Livestock Commission.
Fraser, M.D., R. Fychan, S.T. Evans, M.H.M. Speijers and R. Jones. 1999a. The effect of harvest date and inoculation on the voluntary intake and in vivo digestibility of kale silage by sheep. Proc. Br. Soc. Anim. Sci. p. 96.
Fraser, M.D., M.H.M. Speijers, S.T. Evans and R. Jones. 1999b. Nitrogen utilisation by lambs offered red clover and lucerne silages harvested at two different stages of growth. Proc. Br. Soc. Anim. Sci. p. 95.
Halling, M.A., A. Hopkins, O. Nissinen and U. Sölter. 2000. Production of five forage legumes for silage in northern Europe. Grassld Sci. in Europe 5(in press).
Hatfield, R. and R. Muck. 1999. Characterising proteolytic inhibition in red clover silage. In: Proceedings of the X11 International Silage Conference (T. Pauly, ed), Uppsala, Sweden, pp. 147-148.
Heron, S.J.E., R.A. Edwards and P. McDonald. 1986. Changes in the nitrogenous components of gamma-irradiated and inoculated ensiled grass. J. Sci. Food Agric., 37:979-985.
Hickey, M.W., A.J. Hillier and G.R. Jago. 1983. Peptidase activities in Lactobacilli. Aust. J. Dairy Technol. 38:118-123.
Humphreys, M.O. 1989. Water-soluble carbohydrates in perennial ryegrass breeding. I. Genetic differences among cultivars and hybrid progeny grown as spaced plants. Grass For. Sci. 44:231-236.
Jarvis, S.C., R.J. Wilkins and B.F. Pain. 1996. Opportunities for reducing the environmental impact of dairy farming management: a systems approach. Grass For. Sci. 51:21-31.
Jones, B.A. 1993. Inhibition of proteolysis by red clover. Proc. X Int. Silage Conf., Dublin, pp. 106-107.
Jones, R. 1998. Bridging the protein gap: potential of forage crops for UK livestock production. In: Biotechnology in the Feed Industry, Proc. of Alltech’s 14th Annual Symposium (T.P. Lyons and K.A. Jacques, eds). pp 119-135.
Jones, R., R. Fychan, S. Evans and J. Roberts. 1999. Effects of wilting and application of a bacterial inoculant on the fermentation characteristics of lupin silage. In: Proceedings of the X11 International Silage Conference (T. Pauly, ed), Uppsala, Sweden, pp. 98-99.
Keady, T.W.J. 1998. The production of high feed value grass silage and the choice of compound feed type to maximise animal performance. In: Biotechnology in the Feed Industry, Proc. of Alltech’s 14th Annual Symposium (T.P. Lyons and K.A. Jacques, eds). pp 157-179.
Kemble, A.R. 1956. Studies on the nitrogen metabolism of the ensilage process. J. Sci. Food Agric. 7:125-130.
Lessard, J.L, J.D. Erfle, E.D. Sauer and S. Mahadevan. 1978. Protein and free amino acid patterns in maize ensiled with and without urea. J. Sci. Food Agric. 29:506-5 12.
Mangan J.L., E.A. Harrison and R.L. Vetter. 1991. Immuno-reactive fraction I leaf protein and dry matter content during wilting and ensiling of ryegrass and alfalfa. J. Dairy Sci. 74:2186-2199.
Mansbridge, R.J. and J.S. Blake. 1998. The effect of feeding different protein sources on intake, milk yield, milk composition and liveweight in high yielding Holstein cows. Proc. Br. Soc. Anim. Prod. p. 202.
McDonald, P., A.R. Henderson and S.J.E. Heron. 1991. The Biochemistry of Silage. Second Edn. Chalcombe Publications, Marlow, UK.
Merry, R.J., R. Jones and M.K. Theodorou. 2000. The conservation of grass. In: Grass, Its Production and Utilization (A. Hopkins, ed.), Blackwell Science, Oxford, pp. 196-228.
Messman, M.A., W.P. Weiss and M.E. Koch. 1994. Changes in total and individual proteins during drying, ensiling, and ruminal fermentation of forages. J. Dairy Sci. 77:492-500.
Miller, L.A., M.A. Neville, D.H. Baker, R.T. Evans, M.K. Theodorou, J.C. MacRae, M.O. Humphrys and J.M. Moorby. 1999. Milk production from dairy cows offered perennial ryegrass selected for high water soluble carbohydrate concentrations compared to a control grass. Proc. Br. Soc. Anim. Sci. p. 208.
Moorby, J.M., P.R. Evans and N.E. Young. 1998. Milk production from dairy cows offered a conserved barley/kale bicrop compared to grass silage. Proc. Br. Soc. Anim. Prod. p. 24.
Morrison, J., M.V. Jackson and P.E. Sparrow. 1980. The response of perennial ryegrass to fertiliser nitrogen in relation to climate and soil. Tech. Rep. 27, Grassld Res. Inst., Hurley.
Moss, A.R. and H.F. Grundy. 1996. Lupins: the energy and protein value for ruminants and an evaluation as a protein source for young beef cattle. Occ. Symp. 30, Br. Grassld Soc., pp. 217-220.
Ohshima, M, P. McDonald and T. Acamovic. 1979. Changes during ensilage in the nitrogenous components of fresh and additive-treated ryegrass and lucerne. J. Sci. Food Agric. 30:97-106.
Ohshima, M. and P. McDonald. 1978. A review of the changes in nitrogenous compounds of herbage during ensilage. J. Sci. Food Agric. 29:497- 505.
Pahlow, G., C. Rammer, M. Tuori, and R. Wilkins, 2000. Ensiling of established and novel legumes in Germany, Sweden and Finland. Grassld Sci. in Europe 5 (in press).
Sasaki, M., B.W. Bosman and P.S.T. Tan. 1995. Comparison of proteolytic activities in various Lactobacilli. J. Dairy Res. 62:601-610.
Sharp, R., P.G. Hooper and D.G. Armstrong. 1994. The digestion of grass silages produced using inoculants of lactic acid bacteria. Grass For. Sci. 49:42-53.
Thomson, D.J., D.E. Beever, D.G. Harrison, I.W. Hill and D.F. Osbourn. 1971. The digestion of dried sainfoin and dried lucerne by sheep. Proc. Nutr. Soc. 30, 14A.
Waghorn, G.C. and I.D. Shelton. 1997. Effect of condensed tannins in Lotus corniculatus on the nutritive value of pasture for sheep. J. Agric. Sci. Camb 128:365-372.
Winters, A., J. Lloyd, K. Lowes, R. Jones and R.J. Merry. 1999a. A rapid and economical technique for predicting free amino acid content in legume silage. In: Proceedings of the X11 International Silage Conference (T. Pauly, ed), Uppsala, Sweden, pp. 168-169.
Winters, A., R. Fychan, D.R. Davies, J. Lloyd, R.J. Merry and R. Jones. 1999b. Protein content of a range of ensiled legumes. In: Proceedings of the X11 International Silage Conference (T. Pauly, ed), Uppsala, Sweden, pp. 131-132.
Woodward, S.L., D.A. Clark, P.J. Laboyrie and E.B.L. Jansen. 1999. Effect of Lotus corniculatus and condensed tannins on milk yield and composition of dairy cows. Proc. of the New Zealand Society of Animal Production 59:152-155.
Author: RAYMOND JONES
Institute of Grassland and Environmental Research, Aberystwyth, Wales, UK






.jpg&w=3840&q=75)