Introduction
An excellent livestock management combines good housing, feeding and health. With the increasing demand for animal protein there is need to increase livestock production, hence the need for organic supplements to be added into animal feed. Organic supplements help to amend the existing deficiencies in feed with no harmful effects on human, animal health and the environment. It also plays a vital role in promoting livestock performance, reducing cost, stimulates digestive organs, increase profitability and nutritional value of feed.
Moringa olifera, neem leaf meal, P. longifolia and several other organic supplements have been proven to produce long stable shelf life, improve an animal’s gut health allowing smooth absorption of nutrients from feed subsequently increasing their performance. They are also better alternatives for keeping animals healthy and reduce antibiotics use in animals as they have been widely adopted in many parts of Europe and Asia where the prophylactic use of antibiotics has been banned. According to data from the Antibiotics Stewardship Council, an initiative of the British Poultry Council (BPC) in 2016, the United Kingdom poultry meat sector used 23.72% tons of antibiotics. This corresponds to a 71% reduction in the net use of antibiotics compared to 2012.
Moringa olifera commonly known as drumstick –tree or horseradish tree is a multi-purpose that has given considerable fodder yield in the tropical humid forest zone of Nigeria during wet and dry seasons (Fadyimu et al., 2011). According to Onimisi et al (2007) Moringa olifera can be included up to 30% in rabbit diet without any adverse effect on the growth performance. It can also be included up to 20% in the diets of laying birds without any adverse effect on their production and performance (Kakengi et al. 2007). Moringa leaves meal contains 27.51% crude protein, 19.25% crude fibre, 2.23% crude fat, 7.13% ash, 76.53% moisture, 43.88% carbohydrate and 1296.00 Kj/g calories (Oduro et al., 2008) which makes it an excellent livestock feed including for grass cutters. Moringa olifera is mainly grown for its leaves in Africa and much appreciated for its pods in Asia (Bosch, 2004), its seed powder has antibacterial properties (Aruna and Srilatha, 2012).
Polyalthia longifolia is an evergreen plant commonly used as an ornamental street tree due to its effectiveness in combating noise pollution (Anupam et al, 2008; Prateek et al, 2014). The tree is known to grow over 30ft in height; the leaves of the plant are aromatic and are generally used for decoration. According to Katkar et al (2010), P. longifolia performs several functions such as antibacterial, antifungal, antidiabetic, antitumor, anti-ulcer, antioxidant and used in the treatment of the digestive system. Pharmacological studies on the bark and leaves of the plants shows display effective antimicrobial activity (Faizi et al, 2003), cytotoxic function (Chang et al, 2006; Chen et al, 2006), hypotensive effects (Saleem et al, 2005).
According to Phadnis et al (1998) P.longifoila contains diterpenoids, alkaloids, tannins and mucilage. The seed flour of the plant has been regarded as been under-utilized and with its significant biological and pharmacological activities can be used as additive in diet formulation (Ibironke and Emmanuel, 2014).
Azadirachta indica (Neem) belongs to the family meliaceae is a tropical evergreen tree widely distributed in Asia, Australia, Africa and other parts of the world (Girish and Shankara, 2008; Sombatsiri et al, 1995). A. indicaperforms several functions such as antibacterial, antifungal, antidiabetic, antitumor, anti-ulcer, antioxidant and used in the treatment of the parasitic infections (Subapriya and Nagini, 2005; Biswas et al, 2002). Pharmacological studies on the bark and leaves of the plants shows display effective antimicrobial activity (Chattopadhyay, 1996), the leaves have also been found to contain little amount of proteins and minerals (Ogbuewu et al, 2011).
Wild sunflower (Tithonia diversifolia) is a bushy perennial weed of cultivated crops and road sides. Its leaves are greyish green, the stems are hallow and slightly ridged. T. diversifolia performs several functions such as antibacterial (Victor, 2004), antibacterial (Kareru et al, 2010; Rungeler, 1998), anti-inflammatory (Obafemi et al, 2006), antimicrobial (Liasu et al, 2008), anti-diabetic (Toshihiro et al, 2005) and antioxidant (Robson et al, 2014). According to Akinola et al (1999); Odedire and Oloidi (2014) the plants can be cultivated by resource-poor farmers who could manipulate planting density to achieve maximum yield.
Azolla is a water fern belonging to the family to the family Azollaceae and order pteridophyta, it is rich in essential amino acid, protein, vitamins and minerals like calcium, phosphorus, potassium, magnesium, copper and iron and can be advantageously used for animal fodder. It is a source of nitrogen (Pennerker, 1988), according to Befikadu et al (2008), the chemical composition of Azolla meal are 23.9% crude protein, 13.8% crude fibre, 4.55% ether extract and 10.06% ash.
Grasscutter (Thryonomys swinderianus) also known as cane rat is a wild hystericomorphic rodent widely distributed in the African sub region and exploited in most of areas as a source of animal protein (Asibey, 1974a ; Vos 1978). The colour of the fur is brownish, coarse and thin. Omole et al (2011) also reported that the protein content in the meat is about 20% which compares favourably with other sources of animal protein, the mineral content of the meat contains major minerals such as calcium, phosphorus and iron which are needed for metabolic activities in the body, strong bone formation, regulation of hormonal activities and utilization of other minerals. Grasscutters can utilize high fibrous and cellulose fraction of a feed more than poultry (Alawa, 1993; NRC 1991). Martin (1984) also reported that the animals can adapt to harsh environment and can utilize natural resources and digest most form of edible green stuff ranging from coarse grasses to roughages and household scraps.
Several reports have shown that organic supplements have a stable shelf life, cost effective, naturally enhances performance and add nutritional value to feed, a synergistic combination of Moringa, Azolla, T. diversifolia, P.longifolia and Neem leaf meal would increase profitability, allow the maximization of resources and sustainability on farms. For instance, Abou-Elezz et al (2011) reported that the addition of Moringa leaf meal at 5% significantly increased egg production in laying birds. Alagbe, J. O (2017) reported a significant increase in final live weight of grass cutters fed diet supplemented with 0.5% P. longifolia oil. Esonu et al (2015) reported that the inclusion of Moringa leaf meal at 7.5% in the diet of broiler enhanced their growth process.
Therefore this study was carried out to investigate the effects of Azolla pinnata, Polyalthia longifolia, T. diversifolia, Moringa olifera and Neem leaf infusion as an Organic supplement on the performance and nutrient retention of growing grass cutters.
Materials and Methods
Location of experiment
The experiment was carried out at Dan-malafia Farms, Ogbomosho, Nigeria. The area is located within the derived savannah zone of Nigeria. The research was conducted between September to November, 2017.
Animals and their management
Twenty, 3 months old weaned male grass cutters with an average weight of 580 and 598 grams were randomly assigned to five dietary treatment of four grass cutter per group in a completely randomized experimental design. The grass cutters were housed in a concrete cell measuring 200×80×40cm (length × breath height) which was disinfected one month before the arrival of the animals. The floor of the cell was made of solid concrete, temperature within the cells were within the range of 27-30 during the period of experiment. On introduction of the grass cutters, anti-stress (strexia) was added into the drinking water followed by prophylactic treatment with Ivermectin injection at 0.2ml per animal. Feed intake and weights were recorded daily and weekly respectively throughout the experiment, which lasted for 60 days.
Preparation of experimental diets
Fresh Polyalthia longifolia leaves were harvested from Polyalthia trees in the farm; it is later spread on a concrete floor and allowed to dry for 8 days (air dried). The dried leaves were milled into fine particle size using attrition mill to produce Polyalthia longifolia leaf meal.
Fresh Moringa leaves used for the experimental diets were purchased from Sabo market in Ogbomosho; the leaves were air dried on a concrete floor under shed for 2 weeks. The dried leaves are then hammer milled to produce Moringa leaf meal.
Fresh leaves of Azadirachta indica (Neem) were harvested from matured neem plants within the farm premises and spread out evenly to dry under sunlight for 7 days until the leaves became crispy to torch. The dried leaves were milled to produce neem leaf meal.
Wild sunflower meal was prepared by air drying the wild sunflower strands harvested when the first inflorescence had opened in 50-80% of the plants, it was later spread on a concrete floor to air dry. The dried leaves from the strands were removed and grinded with a hammer mill.
Azolla was collected from ponds within the farm premises and sundried for 6 days, it was later grinded and stored in a container.
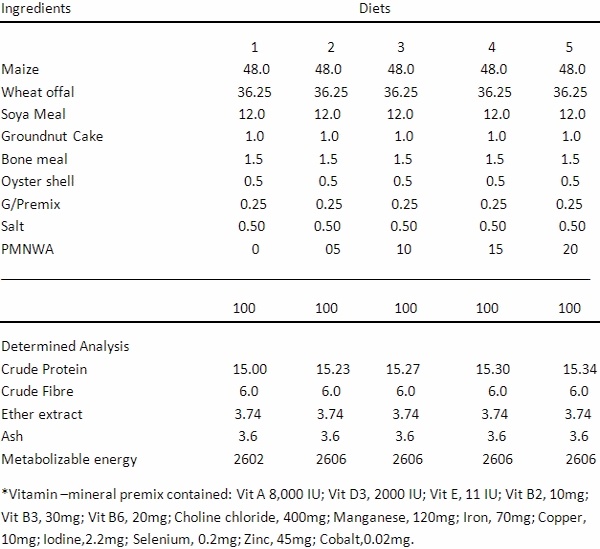
The Polyalthia longifolia leaf meal, Moringa leaf meal, Neem leaf meal, Wild sunflower meal and Azolla meal were mixed together in ratio of 1:1:1:1:1 respectively to form PMNWA. PMNWA was mixed with other ingredients to form five (5) experimental diets as follows:
Treatment 1: 0% inclusion of PMNWA
Treatment 2: Basal diet + 5% level of inclusion of PMNWA
Treatment 3: Basal diet + 10% level of inclusion of PMNWA
Treatment 4: Basal diet + 15% level of inclusion of PMNWA
Treatment 5: Basal diet + 20% level of inclusion of PMNWA
Proximate analysis of Wild sunflower meal, Moringa leaf meal, Neem leaf meal, Azolla meal and P. longifolia leaf meal was carried out as presented in Table 1, 2, 3 4 and 5 respectively.
Nutrient Retention
A nutrient retention trial was carried out at the sixth week of the experiment, a known feed was given to each replicate and their faecal output was collected for 3 days. The faecal samples collected were oven dried at 75*C and grinded before been subjected to chemical analysis.
Cost Analysis
Cost analysis of each diet fed to the experimental birds was calculated. The cost of per kg of ingredient was multiplied with quantity of ingredients to derive the cost per kg.
Chemical Analysis
The proximate components of PMNWA and samples of the five experimental diets were determined by method of A.O.A.C (1990).
Statistical Analysis
All data collected were subjected to one way Analysis of variance (ANOVA) by Steel and Torrie (1980). Significant means separated by Duncan multiple range test (Duncan, 1955).
Table 1. Proximate Composition of Wild Sunflower Leaf Meal.
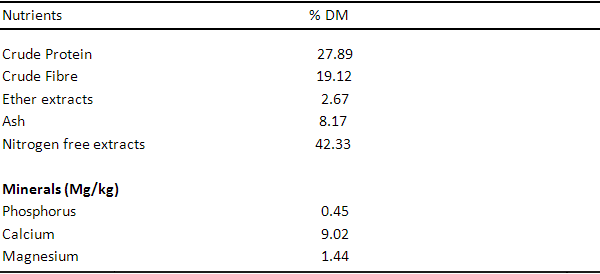
Table 2. Chemical Composition of Moringa Leaf Meal (MLM)
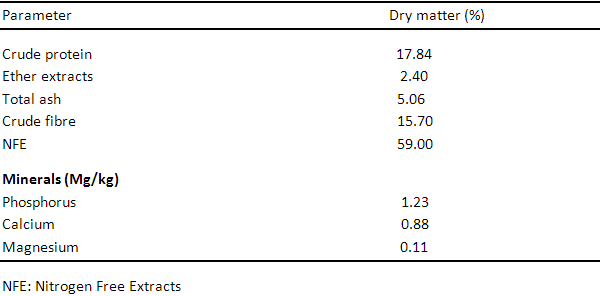
Table 3: Proximate Composition of Neem leaf meal
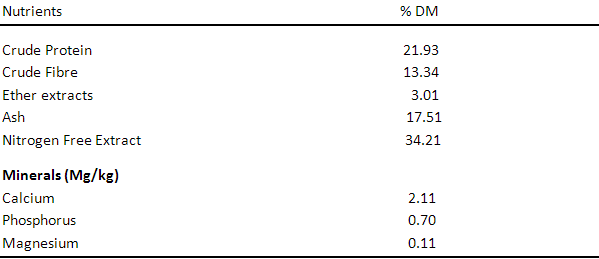
Table 4. Proximate Composition of Azolla Pinnata
Table 5: Proximate composition of P. longifolia leaf meal
Table 6. Percentage Composition of the Experimental Diets
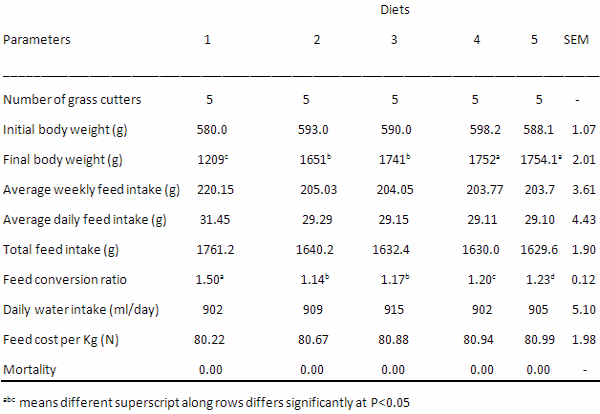
Table 7: Growth Performance of growing grasscutters fed varying levels of PMNWA
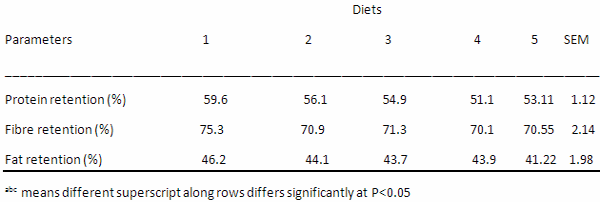
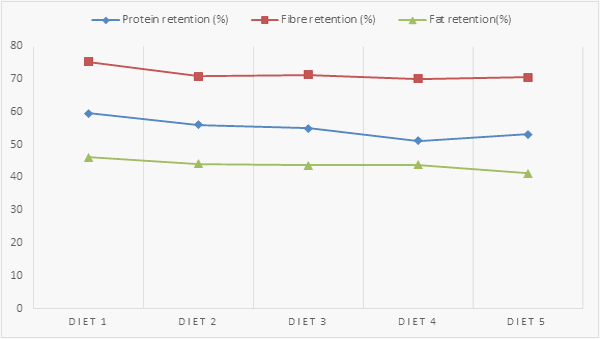
Table 8. Nutrient Retention Parameters of Growing Grasscutters fed varying levels of PMNWA
Figure 1. Performance of growing grasscutters fed different levels of PMNWA
Figure 2. Nutrient retention parameters of growing grasscutters fed different levels of PMNWA
RESULTS
Table 1 shows the proximate analysis of Wild sunflower meal, the proximate components are 20.23%, 10.34%, 7.01%, 15.01%, 46.21%, 2.10mg, 0.90mg and 0.41mg for crude protein, crude fibre, ether extract, ash, nitrogen free extract, phosphorus, calcium and magnesium respectively. Table 3, 4 and 5 show the proximate components of Moringa leaf meal, Neem leaf meal and Azolla respectively. Moringa leaf meal values obtained are 27.89%, 19.12%, 2.67%, 8.17%, 42.33%, 0.45mg, 9.02mg and 1.44 mg for crude protein, crude fibre, ether extract, ash, nitrogen free extract, phosphorus, calcium and magnesium respectively while those of Neem leaf meal are 17.84%, 2.40%, 5.06%, 15.70%, 59.0%, 1.23mg, 0.88mg and 0.11mg for crude protein, crude fibre, ether extract, ash, nitrogen free extract, phosphorus, calcium and magnesium respectively. The proximate component of Azolla used in this study are 21.93%, 13.34%, 3.01%, 17.51%, 34.21%, 2.11mg, 0.70mg and 0.11mg for crude protein, crude fibre, ether extract, ash, nitrogen free extract, phosphorus, calcium and magnesium respectively.
Table 6 shows the nutritional composition of the experimental diets. The crude protein (CP) value increases as the inclusion level of PMNWA increases because of the high proportion of crude protein in the organic supplements (PMNWA) used in this study. However, the values were within the range reported by Wogar (2011) for growing grass cutters.
Table 7 reveals the growth performance traits investigated. The final weight values obtained are 1209.0g, 1651.0g, 1741.0g, 1752.0g and 1754.1g for diets 1, 2, 3, 4 and 5 respectively while the average weekly feed intake values are 220.15, 205.03, 204.05, 203.77 and 203.70 (g) for diets 1, 2, 3, 4 and 5, respectively. The final live weight was significantly (P<0.05) influenced by the different inclusion levels of PMNWA. The total feed intake ranges between 1629.6 g and 1761.2g. The daily water intake values obtained are 902, 909, 915, 902 and 915 (ml/day) for diets 1, 2, 3, 4 and 5 respectively while the feed cost (N) per kg obtained are 80.22, 80.67, 80.88, 80.94 and 80.99 for diets 1, 2, 3, 4 and 5 respectively. The feed cost, water intake and feed intake were not significantly affected (P>0.05) by the dietary inclusion of PMNWA, although, the feed and water intake values slightly increased from diet 1 to 3 before it eventually drops at diet 4 and 5. The feed conversion ratio (FCR) values obtained are 1.50, 1.14, 1.17, 1.20 and 1.23 for diets 1, 2, 3, 4 and 5 respectively. The FCR were significantly (P<0.05) different among the dietary treatments.
The nutrient retention parameters as influenced by the diets are presented on Table 8. The protein retention values obtained are 59.6 %, 56.1%, 54.9%, 51.1% and 53.11% for diets 1, 2, 3, 4 and 5 respectively while the fibre retention values are 75.3, 70.9, 71.3, 70.1 and 70.55 (%)for diets 1, 2, 3, 4 and 5 respectively. The fat retention values obtained are 46.2%, 44.1%, 43.7%, 43.9% and 41.22% for diets 1, 2, 3, 4 and 5 respectively. The protein, fat and fibre retention were not significantly (P>0.05) affected by the dietary inclusion of PMNWA.
DISCUSSION
The experimental diets clearly show that PMNWA is of quality protein, the crude protein contents of the diets increased as the inclusion of PMNWA increases, it significantly (P<0.05) affected the final live weight of the grass cutters in all the treatment groups. This is a clear indication that the inclusion of PMNWA across the treatment can support the growth of the grass cutters, grass cutters fed diet 5 had a better final weight followed by diet 4, 3, 2 and 1 respectively and this could be due to a higher protein content contained in the experimental diet. This agrees with the findings of Odetola et al (2012); Olatunji et al (2016) and Fafiolu et al (2006) when Moringa leaf meals (MLM) and Mango leaf meal were fed to weaner rabbits but contrary to the reports of Hossein Abbasi et al (2015) on the effects of feeding different levels of Sweet orange pulp on the growth performance of broiler chickens and Ojabo et al (2012) on the effect of dried sweet orange fruit peel on the growth performance and haematology of rabbits. Preston and Leng (1987) reported that the growth rate of an animal is determined by the feed intake and digestibility, with feed intake being determined by balance of nutrients especially protein in relation to energy for metabolism. According to Meduna (2002) feeds containing 12-20% crude protein have been reported to be suitable for grass cutters.
According to Omole et al (2012) the quantity of feed consumed by grass cutters depends on the age of the grass cutter, quality of feed, environmental condition, genetic factor and health status. The feed intake of the grass cutters used in this study were not significantly (P>0.05) affected by the PMNWA inclusion, although the feed intake slightly increased from diet 1 after which the values decreased though not at a significant level, this observation shows that PMNWA are well tolerated by the animals. The report is agreement with the findings of Ashong and Brown (2011); Djakalia et al (2011); Abou-Elezz et al (2011) and Nuhu (2010).
The significant (P <0.05) differences in the values obtained FCR across the treatment group agrees with the reports of Olugbemi et al (2010); Melesse et al (2011) and contrary to the findings of Gukaya et al (2014). The synergistic activity of the components in PMNWA helps to reduce disease pressure on the flock, thereby increasing the opportunity for the animals to achieve target weight gain, better FCR (Feed Conversion Ratio) and improved performance.
The data obtained based on the chemical composition of Polyalthia longifolia leaves are similar to the findings of Ojewuyi et al (2014); Esonu et al (2002) and Alagbe, J. O (2017) while those of Moringa olifera leaves agrees with the report of Moyo et al (2011); Murro et al (2003); Oduro et al (2008) and Foidlet et al (2001). The values for all the parameters of Azolla fall within the range reported by Shoukat et al (2015); Alalade and Iyayi (2006) and Querubin et al (1986) but contrary to the report of Basak et al (2002) on the calcium content of Azolla, this difference could be due to species, environment/habitat and method of processing. According to Biplob et al (2002) there are six species of Azolla.
Neem leaf meal analyzed parameters correspond to the reports of Obikaonu et al (2014) but contrary to the reports of Oforjindu (2006) and Esonu et al (2006) while those of Wild sunflower meal is in agreement with the findings of Alagbe, J. O (2016) and Olabanji et al (2007) on the Performance and blood profile of grass cutters fed Wild sunflower meal and studies on haematological and serum biochemical characteristics of weaner rabbits fed different levels of Wild sunflower leaf-blood meal mixture respectively.
Protein, fat, fibre retentions and daily water consumption were not influenced by dietary PMNWA contrary to the reports of Bolu et al (2009) on the effects of feeding graded levels of dried pawpaw seed on the performance, blood profile and carcass evaluation of broilers and Ojabo et al (2012) on the effect of dried sweet orange fruit peel on the growth performance and haematology of rabbits. The water intake is a clear indication that the intestinal walls of the animals are well protected.
No mortality was recorded throughout the experimental period. It has been demonstrated that M. olifera, neem and P. longifolia have multiple biological activities, including antiviral and antibacterial properties attributed mainly to their antioxidant and antiradical activity (Chattopadhyay, 1996; Ogbuewu et al, 2011; Ibironke and Emmanuel, 2016).
CONCLUSION
The results obtained from this study clearly demonstrated that PMNWA could be efficiently utilized and tolerated by grass cutters up to 20% inclusion level without any deleterious effect on performance and health status of growing grass cutters.
RECOMMENDATION
From the results and findings in this study, the following recommendations are made:
- There is need to ascertain the effect of PMNWA on blood profile and carcass characteristics of grass cutters.
- Another study can be carried out using broilers, layers and ruminants to compare the effect of PMNWA on the growth performance traits.
- PMNWA can be produced in large quantity and packaged in 100grm sachets which would only be mixed thoroughly with feed to improve its quality.
- A Similar study can be carried out during the hot season to compare the feed intake, daily water intake, final weight, digestibility and feed conversion ratio.
REFERENCES
Onimisi, P.A., Omage, J.J and Maiguzo, K (2007). Evaluation of Moringa leaf meal as a protein source in rabbit diets, paper presented at the 32nd Annual conference of Nigeria society of Animal production, Calabar March 18-21. Pp 293-294.
Kakengi, A. M. V., Shen, M.N., Sarwart, S.V and Fujihara, T (2007). Can Moringa be used as protein supplement to ruminant diet? Asian – Australian Journal of Animal Science. 18 (1): 42-47
Oduro, I., Ellis, W.O and Owusu, D (2008). Nutritional potential of two leaf vegetables: Moringa olifera and Ipomea batatas leaves, Scientific and Eassy. 3(2): 57-60.
Bosch, C. (2004): Moringa oleifera Lam. Pp 75.
Aruna, M. and Srilatha, N. (2012): Water clarification using Moringa oleifera Lam.seed as a natural coagulant. Current Biotica., 5(4): 472-486.
Anupam, Ghosh, Bidnus, Kanti Das., Soroj, Chatterjee and Goutam Chandra (2008). Antibacterial potentiality and phytochemical analysis of mature leaves of P. longifolia. The South Pacific Journal of Natural Science, Vol 26, 2008.
Prateek, L and Miller, N.D (2014). Antioxidant flavonoids structure function and clinical usage. Alt. Med. Rev. 1:103-111
Katkar, K.V., Suthat, A.C., Chauhan, V.S (2010). Pharmacognosy Review. 2010; 4, 62-68.
Faizi, S., Azher, R. A., Khan, Tauseef, S and Ahmad, A (2003). New antimicrobial alkaloids from the roots of P. longifolia var. pendula. Planta Med. 69-350-5
Chen,C.Y., Chang, F.R., Shih, Y.C., Hsiseh, T. J., Chia Y.C (2006). Cytotoxic constituents of P. longifolia var. pendula: Journal of natural products 2006. 63: 1475-8.
Chang, F.R., Hwang, T.L., Yang, Y.L., C.E Wu. C.C, Issa, H.H., Wu Y.C Planta Medica (2006). 72:1344-1347.
Saleem, R., Ahmed, M., Azeem, M., Khan, R.A and Rasool, N (2005). Hypotensive activity and toxicology of constituents from root bark of P. longifolia var.pendula Phytother Res. 19:881-4.
Phadnis, G., Richard, S.S and Denise, R. F (1998). Lipponcotts illustrated Reviews Biochemistry 3rd ed., Lippincott Williams and Wilkins, Philadelphia. 335-388
Ibironke, Adetolu Ajayi and Emmanuel, Nnamdi Ifedi (2014). Proximate analysis and toxicological studies of P. longifolia seed flour in dietary formulation of albino rats. America Chemical Science Journal. 15(3): 1-12, 2014.
Ogbuewu, I. P., Odoemenam, V. U., Obikaonu, H. O., Opara, M. N., Emenalom, O. O., Uchegbu, M. C., Okoli, I. C ., Esonu, B. O and Iloje, M. U (2011). The growing importance of Neem in Agriculture, Industry, Medicine and Environment: A Review”. Research Jou’l of Medicinal Plants 5(3), 230-245.
Biswas, K. I., Chattopadhyay, R., Banejee, K and U. Bandyspadhyay (2002). Biological activities and medicinal properties of Neem. Curr. Sci. 82(11): 1336-1345.
Chattopadhyay, R. R (1996). Possible mechanism of anti-hyperglycemic effects of Neem leaf extracts. Part IV. General Pharmacology, 27: 431-434.
Girish, D and Shankara, M (2008). Antibody activity and quantitative estimation of azadirachtin and nimbin in Azdirachta indica A. Juss grown in foothills of Nepal Afr. Jou’l Biotech. 8(13): 3084-3091.
Sombatsiri, K., Ermel, K and Schmutterer, H (1995). Other meliaceous plants containing ingredients for integrated pest management and further purpose. In: Schumutter, H (Ed.), the neem tree Azadirachta indica A. Juss and other meliaceous plants. VCH, Germany, pp. 585-597.
Subapriya, M and Nagini, O (2005). Antibacterial effect of Neem Oil on multidrug resistant bacteria isolated from human infections. Int’l Jou’l of Biological & Medical Research, 4(4), 3544-3546.
Olatunji, A.K.., Alagbe, J.O and Hammed, M.A (2016). Effect of varying levels of Moringa olifera meal on performance and blood profile of weaner rabbits. International Journal of Science and Research, ISSN (online): 2319-7064.
Bolu, S.A.O., Sola-Ojo, O.A., Olorunsanya and Idris, K (2009). Effect of graded levels of dried pawpaw seed on performance,haematology, serum biochemistry and carcass evaluation of broilers. International Journal of Poultry Science 8(9):905-909, 2009.
Ojabo, L.D., Adenkola, A.Y and Odaudu, G.I (2012). The effect of dried sweet orange fruit peel meal on the growth performance and haematology of rabbits.
Fafiolu, A.O., Oduguwa, O.O., Bamgbose, A.M., Oso, A.O., Isah, O.A., Olatunji, J.E.N and Jegede, A.V (2006) Feeding value of Mango leaf ( Mangifera indica) for growing rabbits. Journal of Animal and Veterinary Advances 5(10):800-804.
Preston, T.R., Leng, R.A (1987). Matching ruminant production system with available resources in the tropics and sub- tropics. Penambul Books Limited: Armidale NSW, Australia.
Nuhu, F. (2010): Effect of Moringa leaf meal on nutrient digestibility, growth, carcass and
Blood indices of weaner rabbits. Master’s Thesis. Kwame Nkurumah University of Science and Technology. Kumasi, Ghana.
Omole, A.J., Ogunleke, F.O., Kehinde, A.S and Popoola, Y.A (2012). Small and Medium Scale Grass cutter Farming. Green choice Agric Publication. Pages 1-5.
Ashong, J. and Brown, D. (2011): Safety and efficacy of Moringa oleifera powder for growing poultry. J. Anim. Sci., 89: 84.
Djakalia, B., Guichard B, and Soumaila, D. (2011): Effect of Moringa oleifera on growth performance and health status of young post-weaning rabbits. Res. J. Poult. Sci., 4: 7- 11
Abou-Elezz, F., Sarmiento-Franco, L., Santos-Ricalde, R. and Solorio-Sanchez, F. (2011): Nutritional effects of dietary inclusion of Leucaena leucocephala and Moringa oleifera leaf meal on Rhode Island Red hens’ performance. Cub. J. Agri Sci., 45: 163-169.
Olugbemi, T., Mutayoba, S. and Lekule, F. (2010a): Effect of Moringa (Moringa oleifera) inclusion in cassava based diets fed to broilers chickens. Inter. J. Poult. Sci., 9:363- 367
Melesse, A., Tiruneh, W. and Negesse, T. (2011): Effects of feeding Moringa stenopetala leaf meal on nutrient intake and growth performance of Rhode Island Red chicks under tropical climate. Trop Subtrop Agroeco., 14: 485-492.
Gakuya, D., Mbugua, P., Mwaniki, S., Kiama S., Muchemi, G. and Njuguna, A. (2014): Effect of Supplementation of Moringa oleifera (LAM) Leaf Meal in Layer Chicken Feed. Int. J. Poult. Sci., 13 (7): 379-384.
Foidl, N., Makkar, H. and Becker, K. (2001): The potential of Moringa Oleifera for agricultural and industrial uses. Pp 45-76. Food and Agricultural Organization (FAO, 2014): Moringa Traditional crop of the month.
Murro, J., Muhikambele, V. and Sarwatt, S. (2003): Moringa oleifera leaf meal can replace cottonseed cake in the concentrate mix fed with Rhodes grass (Chloris gayana) hay for growing sheep. Livest. Res. Rural Dev., 15(11):234-236.
Moyo, B., Masika, P. Hugo, A. and Muchenje, V. (2011): Nutritional characterization of Moringa (Moringa oleifera Lam.) leaves. African J. Biotech., 10(60): 1292-1293.
Querubin LJ, Alcantara PF and Luis ES (1986). Chemical composition and feeding value of Azolla in broiler ration. Philippines Journal of Veterinary and Animal Science, 13: 46.
Basak B, Pramanik AH, Rahmnan MS, Taradar SU and Roy BC (2002). Azolla (Azolla pinnata) as a feed ingredient in broiler ration. International Journal of Poultry Science, 1: 29-24
Alalade OA and Iyayi EA (2006). Chemical composition and feeding value of Azolla (Azolla pinnata) meal for egg type chicks. International Journal of Poultry Science5: 137-141.
Biplob. B., M.D Ahsan Habib Pramanik., Mohammed Saddiqur Rahman., Sharif Uddin Tarafdar and Bimol Chandra Roy (2002) Azolla as a feed ingredient in broiler ration. Int’l Jou. of Poul. Sci 1(1); 29-34, 2002.
Shoukat, Ara., S. Abdul., M.T Bandry and Manzoor A Khan (2015). Feeding potential of aquatic fern Azolla in broiler chicken. Jou. of Poul. Sci & Tech Pg 15-19.
Alagbe, J.O (2017). Effect of dietary inclusion of Polyalthia longifolia leaf meal compared to antibiotics on the nutrient retention, immune response and serum biochemistry of broiler chickens. Greener Journal of Agricultural Sciences. GJAS, Vol. 7(3), May 2017.
Ojewuyi, O.B., Ajiboye, T.O., Adebanjo, E. O., Balogun, A and Mohammed, A. O (2014) Proximate composition, phytochemical and mineral content of young and mature P. longifolia Sonn. Leaves. Fountain Journal of Natural and Applied Sciences: 2014; 3(1): 10-19
Hossein, Abbasi., Alireza, Seidavi., Wuyi, Liu and Leila, Asadpour (2015). Investigation on the effect of different levels of dried sweet orange pulp on the performance, carcass characteristics, physiological and biochemical parameters of chickens. Saudi Journal of Biological Sciences (2015) 22, 139-146.
Meduna, A.J (2002). Preliminary observations on cane rat (Thryonomys swinderianus, Termminck) feeding and breeding of the Federal College of Forestry, Ibadan. Proceedings of the 27th Annual Conference on Nigerian Society of Animal Production, March 17-21. Federal University of Technology, Akure pp: 304-305.
A.O.A.C (1990). Association of Official Analytical Chemists. Official Methods of Analysis 15th Edition Washington, D.C Pp69-77
Duncan, D.B. (1955). Multiple Range and Multiple F-Test Biometrics 11:1-42
Vos, A.D. (1978) Game as food, a report on its significance in Africa and Latin America Unasylver, (4):2-12. www.inusp.ifo/ajol/journals.jgsa/vol 3no3abs.html
Steel, R. G.D and Torrie, J.H (1980). Principles and procedures of sataistics. A biometrical approach, 2nd edn McGraw-Hill Book Coy. NY. USA.
Alawa, J.P and Umunna, J.P (1993). Alternative feed formulation in the development countries: prospects for utilization of agro-industrial by-products. Journal of Animal Production Research 13(2):63-68.
Asbey, E.O.A (1974). Some ecological and economic aspects of the grass cutters (Thyronomys swinderianus) Mammalian, Rodentia, Hystricomorphal in Ghana Unpbi-Ph.D Thesis of Aberdeen, UK. Pp 305.
Martin, G.H (1984). Bush meat in Nigeria as a natural resource with environmental implications. Environmental Conservations. 10:125-132.
NRC (National Research Council). (1991). Micro- livestock little known small ruminants with a promising economic future. National Academy Press, Washington D.C .Pp 192-282.
Omole, A.J., Ogunleke, F.O., Kehinde, A.S and Popoola, Y.A (2011). Small and medium scale grass cutter farming. Back to Agric series. Green Choice Agric Publications. Pp 1-13, ISBN – 978-978-48110-0-9.
Pannaerker S (1988). Azolla as a livestock and poultry feed. Livestock Adviser, 13: 22-26.
Befikadu B, Pramanik AH, Rahmnan MS, Taradar SU and Roy BC (2008). Azolla (Azolla pinnata) as a feed ingredient in broiler ration. International Journal of Poultry Science, 1: 29-24.
Abou-Elezz, F., Sarmiento-Franco, L., Santos-Ricalde, R. and Solorio-Sanchez, F. (2011): Nutritional effects of dietary inclusion of Leucaena leucocephala and Moringa oleifera leaf meal on Rhode Island Red hens’ performance. Cub. J. Agri Sci., 45:162-168.
Victor B Owoyele, Caleb O. Wuraola et al (2004) Studies on the anti-inflammatory and analgesic properties of Tithonia diversifolia leaf extract. Journal of Ethnopharmacology, Vol 90, Issues 2-3, February 2004, Pages 317-321 / doi:10.1016/j.jep.2003.10.010.
Kareru, P.G, Keriko, J.M, Kenji, G.M, Thiong'o, G.T, Gachanja, A.N, Mukiira, H.N (2010) Antimicrobial activities of skin care preparations from plant extract. African Journal of Traditional, Complementary and Alternative Medicines, Vol. 7, No. 3, 2010, pp. 214-218
Rungeler P, Lyss G et al (1998) Study of three sesquiterpene lactones from Tithonia diversifolia on their anti-inflammatory activity using the transcription factor NF-kappa B and enzymes of the arachidonic acid pathway as targets. Planta Med. 1998 Oct; 64(7):588-93.
C. A Obafemi, T O Sulaimon et al (2006) Antimicrobial activity of extracts and a germacranolidetype sesquiterpene lactone from Tithonia diversifolia leaf extract. African Journal of Biotechnology Vol. 5 (12), pp. 1254-1258, 16 June 2006
Liasu M O and Ayandele A A (2008) Antimicrobial activity of aqueous and ethanolic extracts from Tithonia diversifolia and Bryum coronatum collected from Ogbomoso, Oyo State. Nigeria. Adv. in Nat. Appl. Sci., 2(1): 31-34, 2008.
Toshihiro Mura, Kosuke Nosaka et al (2005) Antidiabetic Effect of Nitobegiku, the Herb Tithonia diversifolia, in KK-Ay Diabetic Mice. Biol. Pharm. Bull. 28(11) 2152—2154 (2005)
Robson Miranda da Gama, Marcelo Guimarães, Luiz Carlos de Abreu, José Armando-Junior (2014). Phytochemical screening and antioxidant activity of ethanol extract of Tithonia diversifolia (Hemsl) A. Gray dry flowers. Asian Pac J Trop Biomed 2014; 4(9): 740-742
Akinola, S., T O Elufloye, O I Alatise et al (2009). Toxicity studies of Tithonia diversifolia A. Gray (Asteraceae) in rats. Journal of Ethnopharmacology, Vol 122, Issue 2, 18 March 2009, Pages 410-415 / doi:10.1016/j.jep.2009.12.007
Odedire, N and Oloidi (2014) Studies of Phytochemical Screening, Acute Toxicity and Anti-Diarrhoeal Effect of Aqueous Extract of Kenyan Tithonia diversifolia Leaves in Rats. British Journal of Pharmacology and Toxicology 3(3): 127-134, 2014.
Mahmoud Aly and Hanan. H Balkhy (2012) .The prevalence of antimicrobial resistance in clinical isolates from gulf corporation council countries. Antimicrobial Resistance and Infection Control 2012 1: 26. https:// doi.org/10.1186/2047-2994-1-26
Alagbe, J.O. (2016). The nutritional evaluation of sweet orange fruit peel meal (SOFPM) as a replacement for maize in the diet of weaner grass cutters. Scholarly Journal of Agricultural Science Vol. 6(8), p. 27-282 December, 2016. Available online at http:/ www.scholarly-journals.com/SJAS

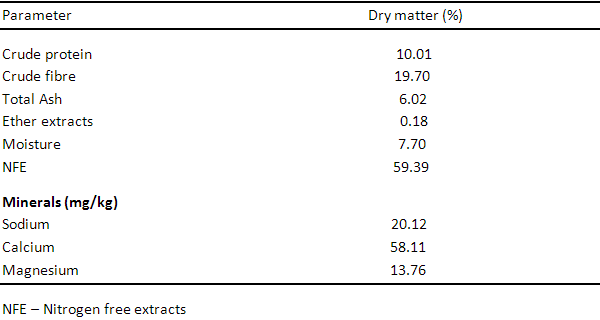








.jpg&w=3840&q=75)