1. Introduction
Mastitis, generally defined as the inflammation of the mammary gland, is a costly and complex disease associated with variable origin, severity and outcome depending on the environment, pathogen and host (De Vliegher et al., 2012; Schukken et al., 2011). Mastitis is caused when pathogenic bacteria enter the sterile environment of the mammary gland, often as a result of disruption of physical barriers such as the teat, requiring prompt and appropriate host defences to prevent colonization and subsequent disease pathology (Aitken et al., 2011; Sordillo et al., 1997). Mastitis causing pathogens are commonly categorized as environmental or contagious, although this distinction has recently been disputed (Zadoks, 2014). Nonetheless, in general environmental pathogens have been grouped to include coliforms like Klebsiella or Escherichia coli (E. coli) and streptococci and are a major cause of clinical mastitis. On the other hand, those categorized as contagious pathogens can readily be spread from the infected quarters to other quarters of the same cow, or other cows and include Staphylococcus aureus (S. aureus) and Streptococcus agalactiae (Atalla et al., 2010b; Bradley, 2002; Contreras and Rodriguez, 2011). Cow factors including age, stage of lactation and somatic cell score history are known to influence the occurrence of mastitis infection (Kehrli, Jr. and Shuster, 1994; Steeneveld et al., 2008). The diverse pathogens that can cause mastitis induce different immune responses in the mammary gland, and therefore the host requires highly specific pathogen-dependent responses for protection (Bannerman, 2009; Wellnitz et al., 2011).
Mastitis infections are described as either subclinical or clinical. Subclinical mastitis is the presence of infection without local inflammation resulting in an absence of visual signs (De Vliegher et al., 2012). It may involve transient cases of inflammation and abnormal milk, and if this persists for longer than two months is termed chronic. Clinical mastitis, on the other hand, is an inflammatory response causing visibly abnormal milk. In the case of mild or moderate clinical mastitis, changes in the udder may include swelling, heat, pain and redness. It is termed severe if the response includes systemic involvement such as fever, anorexia and shock (Ballou, 2011; Zhao and Lacasse, 2008). The diversity as well as the variation in prevalence and abundance of mastitis causing organisms as well as the variation in host responses make mastitis a complex disease that continues to be a burden for the dairy industry.
The bovine mammary gland is equipped with a non-immune anatomical barrier, and a plethora of immune-mediated defense mechanisms that include innate and adaptive immune responses. Innate immunity is relatively non-specific with rapid kinetics while the adaptive immunity offers a highly specific response with relatively delayed kinetics (Borghesi and Milcarek, 2007). Innate host defenses depend on germline-encoded receptors that recognize conserved structures expressed by a wide range of microbes, and early induced cellular and soluble defenses. These natural defenses respond quickly to microbes during early stages of infection and are tightly integrated with the adaptive immune system. The innate host defences of the mammary gland have been reviewed extensively elsewhere (Sordillo and Streicher, 2002; Rainard and Riollet, 2006; Wellnitz and Bruckmaier, 2012). The adaptive immune system uses a diverse repertoire of antigen specific receptors expressed by clonally expanded B and T-lymphocytes to regulate or eliminate the signal elicited by recognition events. Additionally, the induced adaptive immune response has the capacity to establish antigen specific memory for a rapid and augmented response upon subsequent exposure to the same antigen (Iwasak and Medzhitov, 2010). For example, these various components of the immune system work in collaboration both locally and systemically in an attempt to control specific mastitis pathogens invading the mammary gland, but the details of the response is contingent upon the stage of infection and nature of the pathogen, as well as its interaction with the genetics of the host.
The interaction between mastitis pathogens and the host immune system is intricate, since both have the ability to co-evolve to recognize, respond and adapt to the other. As such, microbial pathogens have developed various strategies to alter and evade host defenses in order to survive. Importantly, the host immune system is also adaptive and has a large arsenal to control or eliminate microbial threat. Even so, it is widely accepted that susceptibility of individuals within a given species differs to the same microbial pathogen. This variability in host-pathogen interaction is controlled by the inherent genetic make-up of the host, including innate and adaptive immune responses, particularly the acquired immunological memory, as well as the nature of the microbial pathogen (Hermann, 2007).
Mastitis causing-bacterial pathogens are often well adapted to the bovine host resulting in clinical signs and, occasionally, subclinical infection before they lead to chronicity and persistence in the mammary gland. Persistent intramammary infections are frequently associated with recurrent clinical episodes and long-term increases in milk somatic cells counts. Persistent strains often express sets of genes that relate to their adaptation to the intramammary milieu and allow for intracellular survival and subsequent modulation of host defense mechanisms (Atalla et al 2010a and b). Staphylococcus aureus and E. coli are well studied mastitis pathogens in the context of host-pathogen interaction and the elucidation of their genes, along with host immune response genes, is launching new studies in functional genomics (Hermann, 2007). Understanding sequence data and locating functional SNPs in both the host and pathogen is expected to reveal relationships between immune function and the relevant genes that have the potential to advance resistance to specific pathogens.
Treatment of mastitis is given on the premise that treatment costs will be outweighed by production gains resulting from elimination of infection. Most farms have established mastitis management programs and include strategies such as routine whole herd antibiotic therapy, culling of chronically affected cows, post milking teat disinfection as well as ensuring routine maintenance of milking machines (Bradley, 2002; Zhao and Lacasse, 2008). Due to high treatment costs, lost income due to discarded milk, public health and animal welfare concerns, it would be advantageous for dairy cattle to resist or mount effective immune responses to clear the wide variety of mastitis-causing pathogens. In the case of mastitis the ability to control or tolerate the infection without actually clearing the pathogen, a phenomena known as resilience or tolerance (Bishop and Woolliam, 2014), is not sufficient given that dairy products are consumed by humans and are expected to be free of all potentially harmful pathogens. Antimicrobial treatment has the potential to increase the risk of antibiotic resistant strains of bacteria emerging in the environment (Walsh and Fanning, 2008), although it has been suggested that scientific evidence does not support emerging resistance in pathogens isolated from dairy cows (Oliver et al., 2011). Nonetheless, other non-antibiotic treatment strategies are clearly warranted. Additionally, decreasing the incidence of mastitis would contribute to increased animal welfare as severe signs are associated with pain and discomfort for the cow (Fitzpatrick et al., 2013).
Mastitis is a problem that plagues dairy cattle worldwide; however this review will focus on the mastitis situation in the most economically developed countries. We highlight the genetic regulation of the bovine immune system and its vital contributions to disease resistance, in particular mastitis. Current genetic selection approaches used in the dairy industry to reduce the incidence of disease are reviewed, including the HIR technology; the Immunity+TM sire line, as well as genomics to improve disease resistance or immune response. While the complex interactions of the host and pathogen are fully acknowledged they are only briefly discussed here.
2. Genetic Regulation of the Immune System
Robust, appropriate and timely host defence mechanisms are critical for prompt bacterial clearance and prevention of mastitis and mammary epithelial damage (Zhao and Lacasse, 2008). Bacteria have a large repertoire of virulence factors that are produced at varying concentrations depending on the stage of infection (Bharathan and Mullarky, 2011), and these virulence factors in part determine differences in the magnitude and duration of host immune responses. Further, given the diversity of mastitis-causing pathogens, it is essential for the host to have a broad range of host-defense mechanisms as part of its immunological arsenal. Both innate and adaptive host defences are required to protect the host from infection. Innate defences against mastitis pathogens are rapid and include neutrophil recruitment to the mammary gland to facilitate bacterial clearance through phagocytosis, production of reactive oxygen species, antibacterial peptides, such as lactoferrin and β- lactoglobulin, and defensins, resulting in increases in the somatic cell count (Oviedo-Boyso et al., 2007; Wellnitz and Bruckmaier, 2012). Mammary epithelial cells are known to play a role in early responses through the production of cytokines like IL-8 and other factors with antimicrobial activities (Strandberg et al., 2005, Brenaut et al., 2014). If the bacteria survive these innate host defences, adaptive immune responses mediated by T and B cells are required to clear the infection (Schwarz et al., 2013). The ideal immune response being one that appropriately recognizes epitopes on the invading pathogen to initiate swift and accurate clearance mechanisms while maintaining minimal pathological consequences. In some situations, such as experiments using in vitro or in vivo lipopolysaccaride challenge to measure bovine inflammatory responses, particularly IL-8, have noted that cows with lower IL-8 responses had quicker recovery in terms of somatic cell counts and milk production than those with high IL-8 production (Kandasamy et al., 2011). This may relate to a more moderate inflammatory response generated in these low IL-8 responders. However, it is important to note that this does not mean that cows classified as low responders for other immune response mechanisms, particularly adaptive immune responses are advantageous. In fact, dairy cows classified as high responders (robust and balanced responses) for adaptive immune response traits have been demonstrated to have reduced disease incidence (Mallard et al., 2014). The other thing worth noting in these experiments was the observation that the differences between high and low IL-8 responses seemed to be controlled by epigenetic effects (Green and Kerr, 2014). Epigenetic influences on bovine type 1 (Interferon-γ) and type 2 (IL-4) cytokine production have also been reported in cows classified as high or low responders based on their antibody and cell-mediated immune responses (Paibomesai et al., 2013). Researchers are only beginning to dissect both the genetic and epigenetic mechanisms that control immunity.
Initiation and regulation of adaptive immune responses are critical to the resolution of infection. Cells of the innate immune system recognize conserved pathogen associated molecular patterns from the bacteria by binding pattern recognition receptors on antigen presenting cells (APC) such as macrophages and dendritic cells (Werling et al., 2006). Such pattern recognition receptors include toll-like receptors (TLR) that are located on cell and endosomal membranes (Akira, 2003; Oviedo- Boyso et al., 2007). The association of a TLR with a pathogen associated molecular pattern initiates a downstream signaling cascade leading to the activation of transcription factors, such as NF-κβ, which enter the nucleus, bind target promoters and may induce the production of cytokines and other endogenous mediators. The 10 mammalian TLRs are known to elicit unique responses through intracellular signaling pathways which initiate inflammatory and antimicrobial processes to eliminate the pathogen (Akira, 2003, Reuven et al., 2014). For example, the recognition of lipopolysaccharide (LPS) from Escherichia coli by TLR4, facilitated by additional proteins including CD14, LPS binding protein and myeloid differentiation protein, is associated with production of TNF-α, IL-1β, IL-6 and IL-8. The lipoteichoic acid of Gram positive bacteria like Staphylococcus aureus recognized by TLR2 is associated with only transient increases in TNF-α and IL-1β as well as IgG2 (Oviedo- Boyso et al., 2007). It is well recognized that E coli induces a stronger increase in the pro- inflammatory cytokines TNF-α and IL-1β compared to S. aureus, (Oviedo-Boyso et al., 2007; Petzl et al., 2008; Wellnitz et al., 2011) contributing to the severe clinical signs typically associated with E.coli mastitis as compared to S. aureus where the majority of cases go unnoticed. This draws attention to the fact that although the innate immune responses provide a first line of defence against invading microbial pathogens, including those that cause mastitis, and contours ensuing adaptive immune responses; innate responses have the potential to generate harmful pathology by driving inappropriate or soaring inflammatory cascades (Kandasamy et al., 2011). These need to be carefully considered and closely monitored when considering immunological interventions.
The major histocompatability complex (MHC) plays an essential role in the induction and regulation of immune responses (Ellis and Codner, 2012). The bovine MHC, bovine lymphocyte antigen (BoLA), has been associated with resistance or susceptibility to mastitis (Mallard et al., 1995; Rupp et al., 2007; Sharif et al., 1998; Yoshida et al., 2012), somatic cell count (Chu et al., 2012; Pashmi et al., 2009; Sharif et al., 1998) and immune response (Mallard et al., 1995; Rupp et al., 2007; Thompson-Crispi et al., 2014). Genetic variation, such as single nucleotide polymorphisms (SNP) in other candidate genes associated with resistance or susceptibility to mastitis have been identified, including TLR4 (de Mesquita et al., 2012; Sharma et al., 2008), TLR2 and caspase-recruitment domain 15 (Pant et al., 2008); IL-10 (Verschoor et al., 2009), osteopontin (Alain et al., 2009), IL-8 and its receptor CXCR1 (Galvao et al., 2011; Leyva-Baca et al., 2008; Verbeke et al., 2012), CCL2 and its receptor (Leyva-Baca et al., 2007), as well as a variety of other genes (Pighetti and Elliott, 2011). Other molecules important in host defence against mastitis causing pathogens such as β- defensins have been identified and their complex genetic regulation is beginning to be understood (Meade et al., 2014). The feasibility of breeding for resistance based on one SNP or a combination of SNP depends on the degree of variation each SNP explains in resistance to mastitis. Since mastitis is a complex genetic trait a combination of many genes will ultimately be responsible for resistance to mastitis; however, certain major genes may contribute more benefit than others and it is important that these genes be elucidated.
Recent studies are beginning to uncover information about the epigenetic influences on bovine immune response genes (Karrow et al., 2011). Some studies are now indicating that epigenetic changes are involved in the regulation of type I and II immune responses of mammals (Wilson et al., 2009; Zhu et al., 2010), including cytokine profiles of dairy cows during the peripartum period when the risk of mastitis is the greatest (Paibomesai et al., 2013). Epigenetic modifications have also been demonstrated to play a role in bovine innate immune responses to LPS stimulation (Doherty et al., 2013; Green and Kerr, 2014). Further, microRNA have been found to be differentially expressed upon challenge with mastitis causing pathogens, suggesting a role for microRNA in regulating host responses to mastitis (Jin et al., 2014; Lawless et al., 2013). Indeed, many studies have demonstrated the bovine immune response to be under genetic and epigenetic control, and making use of this information in breeding strategies is anticipated to help improve udder health.
The important question is how to use this information regarding genetic associations with mastitis and the immune system to actually improve disease resistance. This is not necessarily a straight forward question given the plethora of genes, including their additive, dominant, epistatic and epigenetic interactions. It is sometimes possible to make genetic gains in livestock health to a particular disease by selecting for or against a specific gene. Some examples of this include selection against Mareks Disease of poultry based on MHC haplotypes (Schat et al., 1994), bovine dermatopholosis using information on BoLA (Maillard et al., 2003), brachyspina in cattle (Charlier et al., 2012) among others (Charlier et al., 2008). It is generally straightforward to make genetic gains for diseases caused by single recessive disorders, whereas information on single genes or clusters of genes may be less informative when trying to enhance resistance to complex traits, such as mastitis resistance, which is caused by a diverse set of pathogens controlled by a large variety of genes and gene interactions (Rupp and Boichard, 2003).
It is also worth noting that the immune system, which is the body’s main host defence system, is regulated by thousands of genes (Breuer et al., 2013). This points to the critical importance and complex nature of disease resistance as an overall fitness trait (Berry et al., 2011; Gibson and Bishop, 2005). In fact, recent information from a human systems biology data base on immunity known as the Immunogenetic-Related Information Source – IRIS provides evidence for 1,535 immune response genes as of April 2013(http://www.innatedb.com/curatedGenes). This list of genes was curated by IRIS with the following strict definition of a bona fide immune response gene, “a complete gene that produces a functional transcript and demonstrates at least one of the following defence characteristics: (i) Known or putative function in innate or adaptive immunity, (ii) Participates in the development or maturation of immune system components, (iii) Induced by immunomodulators, (iv) Encodes a protein expressed primarily in immune tissues, (v) Participates in an immune pathway that results in the expression of defense molecules, (vi) Produces a protein that interacts directly with pathogens or their products” (http://www.innatedb.com/redirect.do?go=resourcesGeneLists). When a broader definition of immune response genes are given that seeks to retrieve all genes that have some immune system or related functions, such as that provided by the Immunology Database and Analysis Portal (ImmPort), the list of genes is in the range of 6000 (www.immport.org). Although these databases are based on human genes the newest version of the innate immunity database, InnateDB, does incorporate a list of bovine genes, including pathway and molecular interactions (www.innatedb.com). As pointed out by Karin Breuer and colleagues, as the experimental data from cattle research validates genetic interactions and immunological pathways this will allow for a deepened understanding of important bovine diseases, such as mastitis and tuberculosis (Breuer et al., 2013). At the moment, these immunological databases rely largely on orthological-based approach to predict pathways. As of September 2012, the InnateDB contained more than 70,000 bovine interactions based on orthology and pathway analysis could assign to more than 7000 bovine genes (Breuer et al., 2013). However, since the bovine immune system does contain some unique genetic features, such as a novel bovine type 1 interferon family known as IFNX, it will not always suffice to rely on orthogues from other species. Nonetheless, it is interesting to speculate about similar genetic pathways. For example, work in humans has shown that following exposure to bacterial endotoxin a set of 3,714 unique genes were differentially expressed. These changes in genes of interest were confirmed in follow-up microarray experiments (Calvano et al., 2005). Similar transcriptional changes might be predicted in cattle exposed to endotoxin from E. coli following intramammary exposure (Buitenhuis et al., 2011), as the complex plethora of genes involved in response to mastitis, such as that caused by E. coli is well known (Gunther et al., 2009; Mitterhuemer et al., 2010; Jensen et al., 2013). The goal of this type of systems biology research is to provide a portrait of the entire “interactome between the innate and adaptive immune system, as well as its interconnection with other body systems in the hopes to enhance disease prevention and treatment strategies.
3. Genetic Selection for Disease Resistance
Current genetic selection approaches to improve mastitis resistance include both direct and indirect methods. With the exception of Nordic countries that have been selecting for disease resistance for over 35 years (Osteras et al., 2007), most countries breed for mastitis resistance indirectly through somatic cell score (SCS) (Miglior et al., 2005). More recently, France (Govignon-Gion et al., 2012) and Canada (Jamrozik et al., 2013) have launched routine national genetic and genomic evaluations for clinical mastitis. Problems associated with breeding directly for mastitis resistance include low heritability, the need for accurate health recording, and perhaps most importantly, the potential to skew the immune system causing individuals to be susceptible to other harmful pathogens. This skewing is thought to occur since antibody and cell-mediated immune responses are independent or slightly negatively correlated traits (Mouton et al., 1984; Mallard et al., 1992; Thompson-Crispi et al., 2012b; Heriazon et al., 2013). This means that improvement for one of these traits does not translate into improvement of the other adaptive immune response trait. This concept will be discussed in more detail.
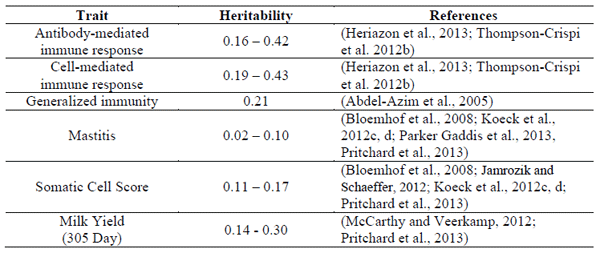
The heritability of mastitis resistance is low, with estimates ranging from about 0.02-0.10 (Koeck et al., 2012c; Parker Gaddis et al., 2014). SCS is genetically correlated (0.7) with mastitis and has a higher heritability of about 0.17 which is why it is used as an alternative trait to breed for resistance to mastitis (Bloemhof et al., 2008; Jamrozik and Schaeffer, 2012; Koeck et al., 2012b). Divergent selection experiments based on SCS in sheep and cattle have been performed with the goal of creating lines of animals with an ability to resist intramammary infection (Brand et al., 2011; Rupp et al., 2009). Although these studies have shown a decrease in mastitis in the low SCS line, caution must be used in this approach to improve udder health. SCS tends to monitor subclinical cases (Barkema et al., 1998) and although decreasing bulk tank counts has been associated with a decline in subclinical mastitis, clinical mastitis continues to be a problem (Olde Riekerink et al., 2008). Further, since the cells that constitute the SCS are cells of the immune system, too low a SCS has been associated with an increased risk of clinical mastitis (Suriyasathaporn et al., 2000). In Canada, the approach will be to equally weight clinical mastitis and SCS in the LPI starting in August 2014. Other immune response traits known to associate with resistance to various diseases, including mastitis, may be added subsequently, although sires with improved immune responses are already available through the Canadian breeding company, the Semex Alliance since December 2012 (Mallard et al., 2014).
In order to select directly for mastitis resistance, accurate disease records are essential. Many countries record disease on a voluntary basis, as is the current situation in the United States (Parker Gaddis et al., 2014; Zwald et al., 2004) and Canada (Koeck et al., 2012a; Neuenschwander et al., 2012). The use of voluntary producer records has brought into question the reliability of the estimates for disease resistance. By applying minimum lactation incidence rates to producer recorded data to include only herds with regular recording, it has been found that although the heritability of disease resistance tends to be low (0.01-0.20) significant genetic variation exists to select for disease resistance (Zwald et al., 2004; Heringstad et al., 2005a; Neuenschwander et al., 2012; Koeck et al., 2012a). Some research has demonstrated the use of genomics to improve the reliability of genetic estimates for disease resistance traits (Parker Gaddis et al., 2014).
Selection against clinical mastitis has the potential to leave cattle susceptible to infection with other mastitis pathogens, since bacteria require unique immune responses for host protection (Schukken et al., 2011), and mastitis pathogens have been demonstrated to change over time and geographically (Bradley, 2002). Further, mastitis causing pathogens tend to be extracellular in nature, requiring robust antibody responses (Thompson-Crispi et al., 2013). Since antibody- and cell-mediated immune responses tend to be negatively genetically correlated (Heriazon et al., 2013; Thompson- Crispi et al., 2012b) selection for mastitis resistance may potentially leave individuals with diminished capability to respond to intracellular pathogens generally controlled by the cell-mediated immune response. Cell-mediated responses have been demonstrated to be critical in controlling Mycobacterium avium spp tuberculosis, the causative pathogen associated with Johne’s disease in cattle (Begg et al., 2011). Maintaining balanced immune responsiveness is an essential consideration in any breeding program to improve animal health. The other contributing factor is that different BoLA alleles have been shown to associate with antibody versus cell-mediated immune responses, as well as mastitis resistance (Rupp et al., 2007). However, these are not the same alleles that associate with resistance to other viral or parasitic pathogens (Stear et al., 2001; Zanotti et al., 1996). Therefore caution must be exercised when selecting for resistance to one specific disease, particularly when it can be caused by multiple pathogens, as is the case with mastitis. Nonetheless, mastitis is such a costly disease it is likely to be included in selection indices in conjunction with other health traits, such as SCS, until alternative approaches based on optimizing host defence mechanisms are more widely available. For example, in Canada an index for Mastitis Resistance was developed that includes both clinical mastitis and SCS traits and will be added to the Lifetime Profitability Index (LPI) in August 2014 (Miglior et al., 2014; Van Doormaal and Beavers, 2014).
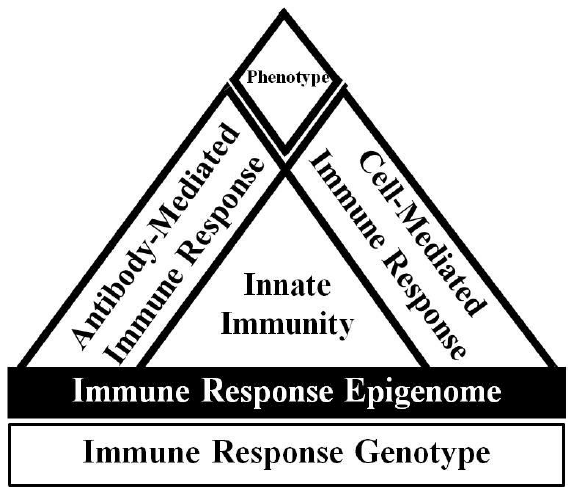
A combination of approaches is likely necessary to decrease mastitis occurrence, such as breeding for broad-based disease resistance based on immune response traits. Breeding for enhanced immune responsiveness is a solution to provide cows with an overall superior ability to respond to a variety of pathogen types requiring unique responses to provide broad-based disease resistance. Individuals with greater and optimally balanced antibody and cell-mediated immune responses breeding values are referred to as High Immune Responders (HIR) (Figure 1) and the method for identifying such individuals is referred to as the HIR technology (Mallard et al., 2014; Wagter-Lesperance et al., 2012).
The HIR technology has been used to identify the ability of cows, calves and bulls to mount antibody and cell-mediated adaptive immune responses (Cartwright et al., 2011; Thompson-Crispi and Mallard, 2012). These adaptive immune response traits are heritable, on average 0.25 – 0.35 (Heriazon et al., 2013; Thompson-Crispi et al., 2012b), considerably higher than estimates for specific clinical or subclinical disease resistance (Table 1). The heritability of immune response is similar to what has been found for milk production traits, indicating it would be possible to make significant genetic gain depending on how heavily health is weighted within the selection index. Cows with superior adaptive immune responses have been demonstrated to have substantially lower occurrence of diseases, including mastitis, metritis, displaced abomasums, retained fetal membranes (Thompson-Crispi et al., 2012a) and are less likely to be seropositive for Mycobacterium avium spp paratuberculosis (Pinedo et al., 2009). It would therefore be feasible and desirable to breed dairy cows for enhanced immune responses to decrease the occurrence of diseases like mastitis (Mallard et al., 2011; Stear et al., 2001). Previously, this approach was shown to improve disease resistance of pigs (Wilkie and Mallard, 1999). It should also be noted that producing robust adaptive immune responses requires appropriate priming via particular innate host defence pathways, such as TLR signalling (Reuven et al., 2014). Priming the immune system with LPS in the udder has been shown to reduce bacterial load in experimentally induced mastitis via the TLR signalling (Gunther et al., 2012; Petzl et al., 2012).
High immune responding cows have also been found to have an increased response to commercial E. coli J5 mastitis vaccination (Wagter et al., 2000) as well as improved colostrum quality as measured by specific antibody (Wagter et al., 2000), total immunoglobulin, lactoferrin and β-lactoglobulin (Fleming et al., 2014). Differences in leukocyte populations between high and low immune responders have also been described, such that cows with superior antibody responses have a higher proportion of B cells in peripheral blood in response to immunization, whereas cows with high cell- mediated responses have a higher baseline proportion of gamma delta (γδ) T cells (Hine et al., 2012). These differences in the diverse phenotypes identified using the HIR technology suggest potential mechanisms that contribute to decreased disease occurrence among high immune responding individuals.
Multiple studies over many years have found beneficial associations between antibody responses and a lower occurrence of mastitis. A study that evaluated antibody-mediated immune responses to a specified test antigen found cows with superior antibody responses had lower occurrence of mastitis in 2 out of 3 herds tested (Wagter et al., 2000). Subsequently, cows with greater antibody responses in a commercial herd in Florida were found to be 1.6-2.5 times less likely to get clinical mastitis compared to other cows in the herd (DeLapaz, 2008; Thompson-Crispi et al., 2012a). Most recently, a nation-wide study in Canada evaluating the incidence rate of clinical mastitis over a 2 year study period found cows with superior antibody responses to have an incidence rate of 17.1 cases of clinical mastitis/100 cow years compared to average and low responding cows with 27.9 and 30.7 cases, respectively. The low responding cows were also found to have more severe mastitis compared to cows with better immune responses (Thompson-Crispi et al., 2013). Antibody-mediated immune responses have also been beneficially genetically correlated with some reproductive traits as well as longevity, suggesting that cows with better immune responsiveness and therefore less disease remain in the herd longer (Thompson-Crispi et al., 2012b).
Conversely, cows with greater cell-mediated immune responses have been found to be less likely to be sero-positive for Mycobacteria avium paratuberculosis (Pinedo et al 2009). Cell-mediated immune responses are also critical to provide protection against Staphylococcus aureus small colony variants that can cause mastitis and have the ability to survive within host cells (Atalla et al., 2010b; Atalla et al., 2010a). Antibody and cell-mediated immune responses have been found to be negatively genetically correlated (Heriazon et al., 2013; Thompson-Crispi et al., 2012b). Consequently, in order to ensure protection to a broad-range of pathogens it is essential to identify and select individuals with the capacity to generate both effective antibody and cell-mediated immune responses (Mallard et al., 2014).
The Semex Alliance utilizes the HIR Technology to identify dairy sires with superior immune responsiveness, termed Immunity+TM.. Daughters of Immunity+TM sires have been found to have lower disease occurrence and higher profitability compared to daughters of sires with either an unknown or an average or low immune response type. Specifically, daughters of Immunity+TM sires in a large herd in the US had a 44% reduction in mastitis, 25% less calf pneumonia and an 8.5% reduction in all diseases in 1st lactation heifers (Mallard et al., 2014). These results highlight the benefit and potential to improve disease resistance, in particular mastitis resistance, by improving overall immune responsiveness.
Genomic selection has allowed for the opportunity to include new phenotypes in breeding objectives, particularly those that may be relatively expensive to measure (Boichard and Brochard, 2012). Genomic selection refers to breeding decisions based on genomic estimated breeding values (GEBV) which are calculated using the joint effects of SNP markers across the entire genome (Schaeffer, 2006; Goddard and Hayes, 2009; Schefers and Weigel, 2012). Using a large reference population with accurate phenotype information, the SNP or haplotype effects for a given trait are estimated. In subsequent generations, only information on the SNP or haplotypes are required to calculate the GEBV (Goddard and Hayes, 2009). Genomic selection has provided many substantial benefits to the dairy industry. Perhaps the most highlighted benefit is in the significant increase in the rate of genetic gain by decreasing generation interval, increasing and selection intensity the accuracy of estimates (Schaeffer, 2006).
The sequencing of the bovine genome and release of SNP arrays used for genomic selection has led to increases in the genome-wide association studies (GWAS). Many GWAS have been performed which has lead to the identification of quantitative trait loci or SNP profiles associated with resistance or susceptibility to mastitis (Sodeland et al., 2011), or somatic cell score as an indicator of mastitis (Meredith et al., 2013; Minozzi et al., 2011; Wijga et al., 2012). Using the approach, many genes involved in immune response have been found, including cytokines IL-4 and IL-13 as well as IL-17 (Lewandowska-Sabat et al., 2012). Recently, a series of GWAS have been performed for general immune responsiveness in dairy cattle and results have been validated in dairy sires (Thompson-Crispi et al., 2014). Results of this work have identified many genes associated with immune responses including the bovine major histocompatability complex, the complement systems as well as cytokines including IL-17 and TNF in the genetic regulation of bovine immune system. Results of these GWAS on mastitis resistance and immune response suggest it is possible to calculate GEBV for mastitis or immune response traits increasing the accuracy of estimates for genetic selection. The next critical steps are to create large reference populations with genotypes and accurate phenotypes for disease and immune response traits in order to improve dairy cattle health.
4. Conclusion
The ideal solutions to improve resistance to mastitis are likely to be those that focus on a large number of genes, by using information from genome-wide association studies, or selection based on breeding values of immune responses which take into account complex genetic interactions between the innate and adaptive host defence mechanisms without the necessity of knowing all about each individual gene. Using selection indices also offers the advantage of being able to easily adjust the weights given to the various traits within the index as the selection proceeds. These two approaches may be best suited to help alleviate mastitis, at least until we gain more knowledge about genetic and epigenetic regulation of host defence mechanisms.
5. Acknowledgement
The authors gratefully acknowledge the agencies that have provided research grants to B.A. Mallard including the Semex Alliance, the Natural Sciences and Engineering Research Council (NSERC), the DairyGen Council of the Canadian Dairy Network (CDN) and the Ontario Ministry of Agriculture, Foods and Rural Affairs (OMAFRA).
6. References
Abdel-Azim, G. A., Freeman, A. E., Kehrli, M. E., Jr., Kelm, S. C., Burton, J. L., Kuck, A. L. et al. (2005). Genetic basis and risk factors for infectious and noninfectious diseases in US Holsteins. I. Estimation of genetic parameters for single diseases and general health. J.Dairy Sci. 88, 1199-1207
Aitken, S. L., Corl, C. M., and Sordillo, L. M. (2011). Immunopathology of mastitis: insights into disease recognition and resolution. J.Mammary.Gland.Biol.Neoplasia. 16, 291-304.
Akira, S. (2003). Mammalian Toll-like receptors. Curr.Opin.Immunol. 15, 5-11.
Alain, K., Karrow, N. A., Thibault, C., St-Pierre, J., Lessard, M., and Bissonnette, N. (2009). Osteopontin: an early innate immune marker of Escherichia coli mastitis harbors genetic polymorphisms with possible links with resistance to mastitis. BMC Genomics. 10, 444.
Atalla, H., Gyles, C., and Mallard, B. (2010a). Persistence of a Staphylococcus aureus small colony variants (S. aureus SCV) within bovine mammary epithelial cells. Vet.Microbiol. 143, 319- 328.
Atalla, H., Wilkie, B., Gyles, C., Leslie, K., Mutharia, L., and Mallard, B. (2010b). Antibody and cell-mediated immune responses to Staphylococcus aureus small colony variants and their 61 parental strains associated with bovine mastitis. Dev.Comp Immunol. 34, 1283-1290.
Ballou, M. A. (2011). Inflammation: Role in the etiology and pathophysiology of clinical mastitis in dairy cows. J.Anim Sci.90, 1466-1478.
Bannerman, D. D. (2009). Pathogen-dependent induction of cytokines and other soluble inflammatory mediators during intramammary infection of dairy cows. J.Anim Sci. 87, 10-25.
Barkema, H. W., Schukken, Y. H., Lam, T. J., Beiboer, M. L., Wilmink, H., Benedictus, G. et al. (1998). Incidence of clinical mastitis in dairy herds grouped in three categories by bulk milk somatic cell counts. J.Dairy Sci. 81, 411-419.
Begg, D. J., de, S. K., Carter, N., Plain, K. M., Purdie, A., and Whittington, R. J. (2011). Does a Th1 over Th2 dominancy really exist in the early stages of Mycobacterium avium subspecies paratuberculosis infections? Immunobiology. 216, 840-846.
Berry, D. P., Bermingham, M. L., Good, M., and More, S. J. (2011). Genetics of animal health and disease in cattle. Ir.Vet.J. 64, 5.
Bharathan, M. and Mullarky, I. K. (2011). Targeting mucosal immunity in the battle to develop a mastitis vaccine. J.Mammary.Gland.Biol.Neoplasia. 16, 409-419.
Bishop, S., and Woolliams, J. A. (2014). Genomics and disease resistance studies in livestock. Livestock Sci. http://dx.doi.org/10.1016/j.livsci.2014.04.034i
Bloemhof, S., de Jong, G., and de Haas, Y. (2008). Genetic parameters for clinical mastitis in the first three lactations of Dutch Holstein cattle. Vet.Microbiol. 134, 165-171.
Boichard, D. and Brochard, M. (2012). New phenotypes for new breeding goals in dairy cattle. Animal. 6, 544-550.
Borghesi, L., and Milcarek, C. (2007). Innate versus adaptive immunity: a paradigm past its prime? Cancer Res. 67:3989-3993.
Bradley, A. (2002). Bovine mastitis: an evolving disease. Vet.J. 164, 116-128.
Brand, B., Hartmann, A., Repsilber, D., Griesbeck-Zilch, B., Wellnitz, O., Kuhn, C. et al. (2011). Comparative expression profiling of E. coli and S. aureus inoculated primary mammary gland cells sampled from cows with different genetic predispositions for somatic cell score. Genet.Sel Evol, 43, 24.
Brenaut, P., Lefevre, L., Rau, A., Laloe, D., Pisoni, G., Moroni, P. et al. (2014). Contribution of mammary epithelial cells to the immune response during early stages of a bacterial infection to Staphylococcus aureus. Vet.Res. 45, 16.
Breuer, K,, Foroushani, A.K., Laird, M.R., Chen, C., Sribnaia, A., Lo, R., Winsor,G.L., Hancock, R.E., Brinkman, F.S., and Lynn, D.J. (2013). InnateDB: systems biology of innate immunity and beyond. Nucleic Acid Res. 41:D1228-1233.
Buitenhuis, B., Røntved, C.M., Edwards, S.M., Ingvartsen, K.L., Sørensen, P. (2011). In depth analysis of genes and pathways of the mammary gland involved in the pathogenesis of bovine Escherichia coli-mastitis. BMC Genomics. 28;12:130.
Calvano, S. E., Xiao, W., Richards, D.R., Felciano, R.M., Baker, H.V., Cho, R.J., Chen, R.O., Brownstein, B.H., Cobb, J.P., Tschoeke, S.K., Miller-Graziano, C., Moldawer, L.L., Mindrinos , M.N., Davis, R.W., Tompkins, R.,G., Lowry, S.F. Inflamm and Host Response to Injury Large Scale Collab. Res. Program. (2005). A network-based analysis of systemic inflammation in humans. Nature. 437, 1032-1037.
Cartwright, S. L., Begley, N., Schaeffer, L. R., Burnside, E. B., and Mallard, B. A. (2011). Antibody and cell-mediated immune responses and survival between Holstein and Norwegian Red x Holstein Canadian calves. J.Dairy Sci. 94, 1576-1585.
Charlier, C., Agerholm, J. S., Coppieters, W., Karlskov-Mortensen, P., Li, W., de, J. G. et al. (2012). A deletion in the bovine FANCI gene compromises fertility by causing fetal death and brachyspina. PLoS.One. 7, e43085.
Charlier, C., Coppieters, W., Rollin, F., Desmecht, D., Agerholm, J. S., Cambisano, N. et al. (2008). Highly effective SNP-based association mapping and management of recessive defects in livestock. Nat.Genet. 40, 449-454.
Chu, M. X., Ye, S. C., Qiao, L., Wang, J. X., Feng, T., Huang, D. W. et al. (2012). Polymorphism of exon 2 of BoLA-DRB3 gene and its relationship with somatic cell score in Beijing Holstein cows. Mol.Biol.Rep. 39, 2909-2914.
Contreras, G. A. and Rodriguez, J. M. (2011). Mastitis: comparative etiology and epidemiology. J.Mammary.Gland.Biol.Neoplasia. 16, 339-356.
de Mesquita, A. Q., CS, E. R., de Mesquita, A. J., Jardim, E. A., and Kipnis, A. P. (2012). Association of TLR4 polymorphisms with subclinical mastitis in Brazilian holsteins. Braz.J.Microbiol. 43, 692-697.
De Vliegher, S., Fox, L. K., Piepers, S., McDougall, S., and Barkema, H. W. (2012). Invited review: Mastitis in dairy heifers: nature of the disease, potential impact, prevention, and control.
J.Dairy Sci, 95, 1025-1040. DeLapaz, J. (2008). Using Humoral and Cellular Response to Novel Antigens in Periparturent Dairy Cows as a Measure of Genetic Disease Resistance in Dairy Cows. MSc Thesis. University of Florida. http://etd.fcla.edu/UF/UFE0022360/delapaz_j.pdf.
Doherty, R., O'Farrelly, C., and Meade, K. G. (2013). Epigenetic regulation of the innate immune response to LPS in bovine peripheral blood mononuclear cells (PBMC). Vet.Immunol.Immunopathol. 154, 102-110
Ellis, S. A. and Codner, G. (2012). The impact of MHC diversity on cattle T cell responses. Vet.Immunol.Immunopathol. 148, 74-77.
Fitzpatrick, C. E., Chapinal, N., Petersson-Wolfe, C. S., Devries, T. J., Kelton, D. F., Duffield, T. F. et al. (2013). The effect of meloxicam on pain sensitivity, rumination time, and clinical signs in dairy cows with endotoxin-induced clinical mastitis. J.Dairy Sci. 96, 2847-2856.
Fleming, K. (2014). Variation of Bioactive Components in Colostrum and Milk from Canadian Holstein Dairy Cattle Classified as High, Average or Low Immune Responders. MSc Thesis. University of Guelph. https://atrium.lib.uoguelph.ca/xmlui/bitstream/handle/10214/8185/Fleming_Kelly_201406_MSc.538 pdf?sequence=3
Galvao, K. N., Pighetti, G. M., Cheong, S. H., Nydam, D. V., and Gilbert, R. O. (2011). Association between interleukin-8 receptor-alpha (CXCR1) polymorphism and disease incidence, production, reproduction, and survival in Holstein cows. J.Dairy Sci. 94, 2083-2091.
Gibson, J. P., and Bishop, S. C. (2005). Use of molecular markers to enhance resistance of livestock to disease: a global approach. Rev.Sci.Tech. 24, 343-353.
Goddard, M. E. and Hayes, B. J. (2009). Mapping genes for complex traits in domestic animals and their use in breeding programmes. Nat.Rev.Genet. 10, 381-391.
Govignon-Gion, A., Dassonneville, R., Baloche, G., Ducrocq, V. (2012). Genetic evaluation of mastitis in dairy cattle in France. Interbull Bulletin No. 46. Cork, Ireland, May 28-31, 2012. Available online https://journal.interbull.org/index.php/ib/article/view/1276/1314.
Green, B.B., Kerr, D.E. (2014). Epigenetic contribution to individual variation in response to lipopolysaccharide in bovine dermal fibroblasts. Vet Immunol Immunopathol.157, 49-58.
Gunther, J., Koczan, D., Yang, W., Nurnberg, G., Repsilber, D., Schuberth, H. J. et al. (2009). Assessment of the immune capacity of mammary epithelial cells: comparison with mammary tissue after challenge with Escherichia coli. Vet.Res. 40, 31.
Gunther, J., Petzl, W., Zerbe, H., Schuberth, H. J., Koczan, D., Goetze, L. et al. (2012). Lipopolysaccharide priming enhances expression of effectors of immune defence while decreasing expression of pro-inflammatory cytokines in mammary epithelia cells from cows. BMC Genomics. 13, 17
Heriazon, A., Quinton, M., Miglior, F., Leslie, K. E., Sears, W., and Mallard, B. A. (2013). Phenotypic and genetic parameters of antibody and delayed-type hypersensitivity responses of lactating Holstein cows. Vet.Immunol.Immunopathol. 154, 83-92.
Hermann, C. (2007). Review: variability of host–pathogen interaction. J Endotoxin Res. 13:199-217.
Hine, B. C., Cartwright, S. L., and Mallard, B. A. (2012). Analysis of leukocyte populations in
Canadian Holsteins classified as high or low immune responders for antibody- or cell- mediated immune response. Can.J.Vet.Res. 76, 149-156.
Iwasaki, A., and Medzhitov, R. (2010). Regulation of adaptive immunity by the innate immune system. Science. 327, 291-295.
Jamrozik, J. and Schaeffer, L. R. (2012). Test-day somatic cell score, fat-to-protein ratio and milk yield as indicator traits for sub-clinical mastitis in dairy cattle. J.Anim Breed.Genet. 129, 11- 19.
Jamrozik, J., Koeck, A., Miglior, F., Kistemaker, G., Schenkel, F., Kelton, D., Van Doormaal, B. (2013). Genetic and Genomic Evaluation of Mastitis Resistance in Canada. Interbull Bulletin No. 47. Nantes, France, August 23-25, 2013. Available online https://journal.interbull.org/index.php/ib/article/view/1291/1365.
Jensen, K., Gunther, J., Talbot, R., Petzl, W., Zerbe, H., Schuberth, H. J. et al. (2013). Escherichia coli- and Staphylococcus aureus-induced mastitis differentially modulate transcriptional responses in neighbouring uninfected bovine mammary gland quarters. BMC Genomics. 14, 36.
Jin, W., Ibeagha-Awemu, E. M., Liang, G., Beaudoin, F., Zhao, X., and Guan, l. L. (2014). Transcriptome microRNA profiling of bovine mammary epithelial cells challenged with Escherichia coli or Staphylococcus aureus bacteria reveals pathogen directed microRNA expression profiles. BMC Genomics. 15, 181.
Kandasamy, S., Green, B.B., Benjamin A.L., Kerr, D.E. (2011) Between-cow variation in dermal fibroblast response to lipopolysaccharide reflected in resolution of inflammation during Escherichia coli mastitis. J. Dairy Sci. 94, 5963-5975.
Karrow, N. A., Sharma, B. S., Fisher, R. E., and Mallard, B. A. (2011). Epigenetics and Animal Health. In Comprehensive Biotechonology, Second Edition (pp. 381-394). Comprehensive Biotechonology Elsevier B.V.
Kehrli, M. E., Jr. and Shuster, D. E. (1994). Factors affecting milk somatic cells and their role in health of the bovine mammary gland. J.Dairy Sci. 77, 619-627.
Koeck, A., Miglior, F., Kelton, D. F., and Schenkel, F. S. (2012a). Health recording in Canadian Holsteins - data and genetic parameters. J.Dairy Sci. 95, 4099-4108.
Koeck, A., Miglior, F., Kelton, D. F., and Schenkel, F. S. (2012b). Alternative somatic cell count traits to improve mastitis resistance in Canadian Holsteins. J.Dairy Sci. 95, 432-439.
Koeck, A., Miglior, F., Kelton, D. F., and Schenkel, F. S. (2012c). Health recording in Canadian Holsteins: data and genetic parameters. J.Dairy Sci. 95, 4099-4108.
Koeck, A., Miglior, F., Kelton, D. F., and Schenkel, F. S. (2012d). Short communication: Genetic parameters for mastitis and its predictors in Canadian Holsteins. J.Dairy Sci. 95, 7363-7366
Lawless, N., Foroushani, A. B., McCabe, M. S., O'Farrelly, C., and Lynn, D. J. (2013). Next generation sequencing reveals the expression of a unique miRNA profile in response to a gram-positive bacterial infection. PLoS.One. 8, e57543.
Lewandowska-Sabat, A. M., Gunther, J., Seyfert, H. M., and Olsaker, I. (2012). Combining quantitative trait loci and heterogeneous microarray data analyses reveals putative candidate pathways affecting mastitis in cattle. Anim Genet. 43, 793-799.
Leyva-Baca, I., Schenkel, F., Martin, J., and Karrow, N. A. (2008). Polymorphisms in the 5' upstream region of the CXCR1 chemokine receptor gene, and their association with somatic cell score in Holstein cattle in Canada. J.Dairy Sci. 91, 407-417.
Leyva-Baca, I., Schenkel, F., Sharma, B. S., Jansen, G. B., and Karrow, N. A. (2007). Identification of single nucleotide polymorphisms in the bovine CCL2, IL8, CCR2 and IL8RA genes and their association with health and production in Canadian Holsteins. Anim Genet. 38, 198-202.
Maillard, J. C., Berthier, D., Chantal, I., Thevenon, S., Sidibe, I., Stachurski, F. et al. (2003). Selection assisted by a BoLA-DR/DQ haplotype against susceptibility to bovine dermatophilosis. Genet.Sel Evol. 35 Suppl 1, S193-S200.
Mallard, B.A., Wilkie, B.N., Kennedy, B.W., Quinton, M. (1992). Use of estimated breeding values in a selection index to breed Yorkshire pigs for high and low immune and innate resistance factors. Anim. Biotechnol. 3, 257-280
Mallard, B. A., Leslie, K. E., Dekkers, J. C., Hedge, R., Bauman, M., and Stear, M. J. (1995). Differences in bovine lymphocyte antigen associations between immune responsiveness and risk of disease following intramammary infection with Staphylococcus aureus. J.Dairy Sci. 78, 1937-1944.
Mallard, B. A., Atalla, H., Cartwright, S., Hine, B. C., Hussey, B., Paibomesai, M. et al. (2011). Genetic and Epigenetic Regulation of the Bovine Immune System: Practical Implications of the High Immune Response Technology. Proc.National Mastitis Council 50th Annual Meeting, 53-63.
Mallard, B. A., Cartwright, S., Emam, M., Fleming, K., Gallo, N., Hodgins, D. C. et al. (2014). Genetic Selection of Cattle for Improved Immunity and Health. In - Advances in Dairy Technology: Strategies to Optimize Performance. Proc Western Canadian Dairy Seminar. 26, 247-257.
McCarthy, J. and Veerkamp, R. F. (2012). Estimation of genetic parameters for test-day records of dairy traits in a seasonal calving system. J.Dairy Sci. 95, 5365-5377
Meade, K. G., Cormican, P., Narciandi, F., Lloyd, A., and O'Farrelly, C. (2014). Bovine beta- defensin gene family: opportunities to improve animal health? Physiol Genomics. 46, 17-28.
Meredith, B. K., Berry, D. P., Kearney, F., Finlay, E. K., Fahey, A. G., Bradley, D. G. et al. (2013). A genome-wide association study for somatic cell score using the Illumina high-density bovine beadchip identifies several novel QTL potentially related to mastitis susceptibility. Front Genet. 4, 229.
Miglior, F., Muir, B. L., and Van Doormaal, B. J. (2005). Selection indices in Holstein cattle of various countries. J.Dairy Sci. 88, 1255-1263.
Miglior, F., Koeck, A., Jamrozik, J., Schenkel, F.S., Kelton, D.F., Kistemaker, G.J., Van Doormaal, B.J. Index for Mastitis Resistance and Use of BHBA for Evaluation of Health Traits in Canadian Holsteins. Interbull Bulletin No. 48. Berlin, Germany, May 20 - 21, 2014. Available online https://journal.interbull.org/index.php/ib/article/view/1349/1420.
Minozzi, G., Nicolazzi, E. L., Strozzi, F., Stella, A., Negrini, R., jmone-Marsan, P. et al. (2011). Genome wide scan for somatic cell counts in holstein bulls. BMC.Proc. Suppl 4, S17.
Mitterhuemer, S., Petzl, W., Krebs, S., Mehne, D., Klanner, A., Wolf, E. et al. (2010). Escherichia coli infection induces distinct local and systemic transcriptome responses in the mammary gland. BMC Genomics. 11, 138.
Mouton, D., Bouthillier,Y., Mevel,J.C., Biozzi,G. (1984). Genetic selection for antibody responsiveness in mice: further evidence for inverse modification of macrophage catabolic activity without alteration of the expression of T-cell-mediated immunity. Ann. Immunol. (Paris) 135D, 173-186.
Mulder, H. A., Lidauer, M. H., Vilkki, J. H., Stranden, I., and Veerkamp, R. F. (2011). Marker- assisted breeding value estimation for mastitis resistance in Finnish Ayrshire cattle. J.Dairy Sci. 94, 4164-4173.
Neuenschwander, T. F., Miglior, F., Jamrozik, J., Berke, O., Kelton, D. F., and Schaeffer, L. R. (2012). Genetic parameters for producer-recorded health data in Canadian Holstein cattle. Animal. 6, 571-578.
Olde Riekerink, R. G., Barkema, H. W., Kelton, D. F., and Scholl, D. T. (2008). Incidence rate of clinical mastitis on Canadian dairy farms. J.Dairy Sci. 91, 1366-1377.
Oliver, S. P., Murinda, S. E., and Jayarao, B. M. (2011). Impact of antibiotic use in adult dairy cows on antimicrobial resistance of veterinary and human pathogens: a comprehensive review. Foodborne.Pathog.Dis. 8, 337-355.
Osteras, O., Solbu, H., Refsdal, A. O., Roalkvam, T., Filseth, O., and Minsaas, A. (2007). Results and evaluation of thirty years of health recordings in the Norwegian dairy cattle population. J.Dairy Sci. 90, 4483-4497.
Oviedo-Boyso, J., Valdez-Alarcon, J. J., Cajero-Juarez, M., Ochoa-Zarzosa, A., Lopez-Meza, J. E., Bravo-Patino, A. et al. (2007). Innate immune response of bovine mammary gland to pathogenic bacteria responsible for mastitis. J.Infect. 54, 399-409.
Paibomesai, M., Hussey, B., Nino-Soto, M., and Mallard, B. A. (2013). Effects of parturition and dexamethasone on DNA methylation patterns of IFN-gamma and IL-4 promoters in CD4+ T- lymphocytes of Holstein dairy cows. Can.J.Vet.Res. 77, 54-62.
Pant, S. D., Schenkel, F. S., Leyva-Baca, I., Sharma, B. S., and Karrow, N. A. (2008). Identification of polymorphisms in bovine TLR2 and CARD15,associations between CARD15 polymorphisms and milk somatic cell score in Canadian Holsteins, and functional relevance of SNP c.3020A>T. Dev.Biol.(Basel). 132, 247-253.
Parker Gaddis, K. L., Cole, J. B., Clay, J. S., and Maltecca, C. (2014). Genomic selection for producer-recorded health event data in US dairy cattle. J.Dairy Sci. 97, 3190-3199.
Pashmi, M., Qanbari, S., Ghorashi, S. A., Sharifi, A. R., and Simianer, H. (2009). Analysis of relationship between bovine lymphocyte antigen DRB3.2 alleles, somatic cell count and milk traits in Iranian Holstein population. J.Anim Breed.Genet. 126, 296-303.
Petzl, W., Zerbe, H., Gunther, J., Yang, W., Seyfert, H. M., Nurnberg, G. et al. (2008). Escherichia coli, but not Staphylococcus aureus triggers an early increased expression of factors contributing to the innate immune defense in the udder of the cow. Vet.Res. 39, 18.
Petzl, W., Gunther, J., Pfister, T., Sauter-Louis, C., Goetze, L., von, A. S. et al. (2012). Lipopolysaccharide pretreatment of the udder protects against experimental Escherichia coli mastitis. Innate.Immun.18, 467-477.
Pighetti, G. M. and Elliott, A. A. (2011). Gene polymorphisms: the keys for marker assisted selection and unraveling core regulatory pathways for mastitis resistance. J.Mammary.Gland.Biol.Neoplasia. 16, 421-432.
Pinedo, P. J., Donovan, A., Rae, O., and DeLapaz, J. (2009). Association between paratuberculosis infection and general immune status in dairy cattle. Proceedings of the 10th International Colloquium on Paratuberculosis, 1, 127.
Pritchard, T., Coffey, M., Mrode, R., and Wall, E. (2013). Genetic parameters for production, health, fertility and longevity traits in dairy cows. Animal. 7, 34-46
Rainard, P., and Riollet, C. (2006). Innate immunity of the bovine mammary gland. Vet.Res. 37, 369- 400.
Reuven, E.M., Fink, A., and Shai, Y. (2014). Regulation of innate immune responses by transmembrane interactions: lessons from the TLR family. Biochim Biophys Acta. 1838:1586- 93.
Rupp, R., and Boichard, D. (2003). Genetics of resistance to mastitis in dairy cattle. Vet.Res. 34, 671- 688.
Rupp, R., Hernandez, A., and Mallard, B. A. (2007). Association of bovine leukocyte antigen (BoLA) DRB3.2 with immune response, mastitis, and production and type traits in Canadian Holsteins. J.Dairy Sci. 90, 1029-1038.
Rupp, R., Bergonier, D., Dion, S., Hygonenq, M. C., Aurel, M. R., Robert-Granie, C. et al. (2009). Response to somatic cell count-based selection for mastitis resistance in a divergent selection experiment in sheep. J.Dairy Sci. 92, 1203-1219.
Schaeffer, L. R. (2006). Strategy for applying genome-wide selection in dairy cattle. J.Anim Breed.Genet. 123, 218-223.
Schat, K. A., Taylor, R. L., Jr., and Briles, W. E. (1994). Resistance to Marek's disease in chickens with recombinant haplotypes to the major histocompatibility (B) complex. Poult.Sci. 73, 502- 508.
Schefers, J. M. and Weigel, K. A. (2012). Genomic selection in dairy cattle: Integration of DNA testing into breeding programs. Animal Frontiers. 2, 4-9.
Schukken, Y. H., Gunther, J., Fitzpatrick, J., Fontaine, M. C., Goetze, L., Holst, O. et al. (2011). Host-response patterns of intramammary infections in dairy cows. Vet.Immunol.Immunopathol. 144, 270-289.
Schwarz, D., Rivas, A. L., Konig, S., Diesterbeck, U. S., Schlez, K., Zschock, M. et al. (2013). CD2/CD21 index: a new marker to evaluate udder health in dairy cows. J.Dairy Sci. 96, 5106-5119.
Sharif, S., Mallard, B. A., Wilkie, B. N., Sargeant, J. M., Scott, H. M., Dekkers, J. C. et al. (1998). Associations of the bovine major histocompatibility complex DRB3 (BoLA-DRB3) alleles with occurrence of disease and milk somatic cell score in Canadian dairy cattle. Anim Genet. 29, 185-193.
Sharma, B. S., Mount, J., and Karrow, N. A. (2008). Functional characterization of a single nucleotide polymorphism in the 5' UTR region of the bovine toll-like receptor 4 gene. Dev.Biol.(Basel). 132, 331-336.
Sodeland, M., Kent, M. P., Olsen, H. G., Opsal, M. A., Svendsen, M., Sehested, E. et al. (2011). Quantitative trait loci for clinical mastitis on chromosomes 2, 6, 14 and 20 in Norwegian Red cattle. Anim Genet. 42, 457-465.
Sordillo, L. M., Shafer-Weaver, K., and DeRosa, D. (1997). Immunobiology of the mammary gland. J.Dairy Sci. 80, 1851-1865.
Sordillo, L. M., and Streicher, K. L. (2002). Mammary gland immunity and mastitis susceptibility. J.Mammary.Gland.Biol.Neoplasia. 7, 135-146.
Stear, M. J., Bishop, S. C., Mallard, B. A., and Raadsma, H. (2001). The sustainability, feasibility and desirability of breeding livestock for disease resistance. Res.Vet.Sci. 71, 1-7.
Steeneveld, W., Hogeveen, H., Barkema, H. W., van den, B. J., and Huirne, R. B. (2008). The influence of cow factors on the incidence of clinical mastitis in dairy cows. J.Dairy Sci. 91, 1391-1402.
Strandberg, Y., Gray, C., Vuocolo, T., Donaldson, L., Broadway, M., and Tellam, R. (2005). Lipopolysaccharide and lipoteichoic acid induce different innate immune responses in bovine mammary epithelial cells. Cytokine. 31, 72-86.
Suriyasathaporn, W., Schukken, Y. H., Nielen, M., and Brand, A. (2000). Low somatic cell count: a risk factor for subsequent clinical mastitis in a dairy herd. J.Dairy Sci. 83, 1248-1255.
Thompson-Crispi, K. A. and Mallard, B. A. (2012). Type 1 and Type 2 Immune Response Profiles of Commercial Dairy Cows in Four Regions of Canada. Can.J.Vet.Res. 76, 120-128.
Thompson-Crispi, K. A., Hine, B., Quinton, M., Miglior, F., and Mallard, B. A. (2012a). Short communication: Association of disease incidence and adaptive immune response in Holstein dairy cows. J.Dairy Sci. 95, 3888-3893.
Thompson-Crispi, K. A., Sewalem, A., Miglior, F., and Mallard, B. (2012b). Genetic Parameters of Adaptive Immune Response Traits in Canadian Holsteins. J.Dairy Sci. 95, 401-409.
Thompson-Crispi, K. A., Miglior, F., and Mallard, B. A. (2013). Incidence Rates of Clinical Mastitis among Canadian Holsteins Classified as High, Average and Low Immune Responders. Clin.Vaccine Immunol. 20, 106-112.
Thompson-Crispi, K. A., Sargolzaei, M., Ventura, R., Abo-Ismail, M., Miglior, F., Schenkel, F. et al. (2014). A Genome-Wide Association Study for Immune Response Traits in Canadian Holstein Cattle. BMC Genomics. 15, 559-569.
Van Doormaal, B., Beavers, L. (2014). Mastitis Resistance Selection: Now a Reality! http://www.cdn.ca/articles.php
Verbeke, J., Piepers, S., Peelman, L., Van, P. M., and De, V. S. (2012). Pathogen-group specific association between CXCR1 polymorphisms and subclinical mastitis in dairy heifers. J.Dairy 761 Res. 79, 341-351.
Verschoor, C. P., Pant, S. D., Schenkel, F. S., Sharma, B. S., and Karrow, N. A. (2009). SNPs in the bovine IL-10 receptor are associated with somatic cell score in Canadian dairy bulls. Mamm.Genome. 20, 447-454.
Wagter, L. C., Mallard, B. A., Wilkie, B. N., Leslie, K. E., Boettcher, P. J., and Dekkers, J. C. (2000). A quantitative approach to classifying Holstein cows based on antibody responsiveness and its relationship to peripartum mastitis occurrence. J.Dairy Sci. 83, 488-768 498.
Wagter-Lesperance, L., Cartwright, S., Funk, T., Kelton, D., Miglior, F., and Mallard, B. (2012). Feasibility of high immune response (HIR) technology as a health management tool to characterize immune response profiles of dairy cattle. In H.Hogeveen and T. J. Lam (Eds.), Udder Health and Communication (pp. 359-366). Wageningen Academic Publishers.
Walsh, C. and Fanning, S. (2008). Antimicrobial resistance in foodborne pathogens--a cause for concern? Curr.Drug Targets. 9, 808-815.
Wellnitz, O., Arnold, E. T., and Bruckmaier, R. M. (2011). Lipopolysaccharide and lipoteichoic acid induce different immune responses in the bovine mammary gland. J.Dairy Sci. 94, 5405-5412.
Wellnitz, O. and Bruckmaier, R. M. (2012). The innate immune response of the bovine mammary gland to bacterial infection. Vet.J. 192, 148-152.
Werling, D., Piercy, J., and Coffey, T. J. (2006). Expression of TOLL-like receptors (TLR) by bovine antigen-presenting cells-potential role in pathogen discrimination? Vet.Immunol.Immunopathol. 112, 2-11.
Wijga, S., Bastiaansen, J. W., Wall, E., Strandberg, E., de, H. Y., Giblin, L. et al. (2012). Genomic associations with somatic cell score in first-lactation Holstein cows. J.Dairy Sci. 95, 899-908.
Wilkie, B. and Mallard, B. (1999). Selection for high immune response: an alternative approach to animal health maintenance? Vet.Immunol.Immunopathol. 72, 231-235.
Wilson, C. B., Rowell, E., and Sekimata, M. (2009). Epigenetic control of T-helper-cell differentiation. Nat.Rev.Immunol. 9, 91-105.
Yoshida, T., Furuta, H., Kondo, Y., and Mukoyama, H. (2012). Association of BoLA-DRB3 alleles with mastitis resistance and susceptibility in Japanese Holstein cows. Anim Sci.J. 83, 359- 366.
Zadoks, R. (2014).Understanding the sources, transmission routes and prognoses for mastitis pathogens. In - Advances in Dairy Technology: Strategies to Optimize Performance. Proc Western Canadian Dairy Seminar. 26, 91-100.
Zhao, X. and Lacasse, P. (2008). Mammary tissue damage during bovine mastitis: causes and control. J.Anim Sci. 86, 57-65.
Zhu, J., Yamane, H., and Paul, W. E. (2010). Differentiation of effector CD4 T cell populations (*). Annu.Rev.Immunol. 28, 445-489.
Zwald, N. R., Weigel, K. A., Chang, Y. M., Welper, R. D., and Clay, J. S. (2004). Genetic selection for health traits using producer-recorded data. I. Incidence rates, heritability estimates, and sire breeding values. J.Dairy Sci. 87, 4287-4294.
Table 1. Heritability estimates of immune response, mastitis resistance and milk production and in Holstein dairy cattle
Figure 1. Overview of immune response adapted from Wilkie and Mallard, 1999. The host immune response phenotype is ultimately determined by the interaction of the immune response genotype with the environment. The expression of the immune response genotype is also regulated by epigenetic effects. The innate immune response is relatively fast acting and non- specific, but is critical to signal appropriate adaptive cell-mediated and antibody-mediated immune responses. Dairy cattle with enhanced and balanced cell and antibody-mediated immune responses are known as High Immune Responders.
This article first appeared published in Frontiers to Immunology, September 2014.