The US rainbow trout industry: challenges and opportunities
The Idaho industry
Rainbow trout production in Idaho is estimated at 40-50 million lbs (20 mT) annually, generating revenues of over $70-90 million per year. This accounts for 70-80% of the rainbow trout produced as food fish in the US. In Idaho, the first commercial trout farm was started in the early 1900s, near Twin Falls. There are currently about 120 commercial trout rearing facilities and four state and federal hatcheries in the mid-Snake River area making it the most intensive freshwater salmonid aquaculture area in North America.
Rainbow trout aquaculture sites are supplied with abundant, constant temperature (15°C) spring water from the Snake River Plain aquifer, which contains about 200 billion cubic feet (56.6 million cubic meters) of water (Maley, 1987). The commercial rainbow trout industry primarily occurs within a 94 mile middle section of the Snake River (Figure 1). Spring water flows by gravity through a series of raceways with four to six raceways receiving water in series. The raceways are generally made of concrete, are 20 to 40 m long, 2.5 to 6 m wide and have water depths of 0.8 to 1.2 m (Figure 2). It is estimated that about 11,800 kg of intensively cultured rainbow trout are produced in each raceway using approximately 1700 L/min of water flow on an annual basis. Water use is non-consumptive since 100% of the water is passed directly to the Snake River.
THE US RAINBOW TROUT INDUSTRY IN THE GLOBAL MARKETPLACE
The most important challenge to the Idaho rainbow trout industry along with other domestic farm-raised finfish is increasing international competition from imported farm-raised seafood. The US Department of Commerce estimates that 75% of all seafood consumed in the US is imported; and this percentage is likely to increase with time unless domestic producers can become more price competitive. A large percentage of that increase is farm-raised species. Most, if not all, of these species are raised in countries without the environmental stewardship expectations or requirements that exist in the US, which have a significant impact on costs of production. Consequently, producers in those countries have production costs significantly lower than those in the domestic market. In the past 10 years, global aquaculture production has increased nearly 60%. Internationally, Atlantic salmon production increased 75%, tilapia production increased 70%, rainbow trout production increased 33% and shrimp production increased about 20%. Along with the increase in global aquaculture production has been an increase in the import of the same species. In the past 10 years, shrimp imports in the US increased almost 25%, rainbow trout imports have increased 36% (Figure 3), salmon imports increased 88% and tilapia imports increased 95%. For 2002, the value of imported shrimp, trout, Atlantic salmon and tilapia totaled slightly more than $4.6 billion. For domestic aquatic animal producers these imports have had a significant negative impact on farm gate prices.
Figure 1.Rainbow trout production in Idaho primarily occurs within the middle section of the Snake River. This 94 mile (151 km) stretch extends from Milner Dam at Burley to Bliss Dam, below Hagerman at Bliss.

Figure 2.In Idaho, rainbow trout are generally produced in concrete raceways where spring water flows by gravity through a series of four to six raceways before leaving a production facility.
Nutrition and water quality
With the passage of the 1972 Clean Water Act, National Pollutant Discharge Elimination Permits (NPDES) were issued for all trout farms producing 20,000 lbs. (9,072 kg) or more fish per year. Since 1979, NPDES permits have limited total suspended solids (TSS) discharged to a mean of 5 mg/L, and total settleable solids to 0.1 mg/L. It should be noted that no other agricultural activity in the mid-Snake River area, where the Idaho rainbow trout industry is located, is required to have a discharge permit.
Because of the mandated monthly average effluent TSS limitations, many raceways were redesigned to include a settling basin or quiescent zone at the end of each raceway. The purpose of the settling basin is to efficiently capture solids. Captured solids are removed from the settling basin weekly by pumping or siphoning into a bioresiduals collection pond. This pond is cleaned at least monthly, depending upon loading. Removed bioresiduals are transported out of the Snake River Canyon and distributed as fertilizer onto agricultural land. Good management practices, fish health, and cost effective production each benefit from efficient bioresiduals collection.
In the US, regulation of water quality is overseen by a federal agency (Environmental Protection Agency), but responsibility for monitoring and compliance is held by individual states. Decisions concerning the effect of fish farms on the freshwater environment are, in part, dependent on two factors: (1) nutrient loads in effluent water; and (2) the quality of the stream, river or lake into which the effluent is discharged. After four years of development, Idaho became the first state in the US to implement more strict NPDES permit regulations for fish farms in September, 1999 (Table 1). Additionally, as part of the new NPDES permit, an Effluent Characterization Study was to be conducted during the first 18 months after receiving authorization to discharge under the new permit. Those receiving permits were required to complete the study, which assessed the concentrations and loads of selected discharged pollutants for a 12 month consecutive period (Table 2).
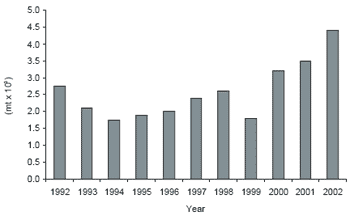
Figure 3.Trout imports to the US, 1992-2002 (National Marine Fisheries Service, 2003).
Table 1.NPDES permit effluent limits and monitoring requirements for Idaho aquaculture facilities.
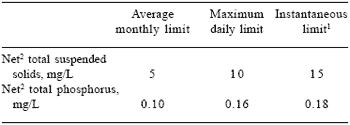
1Instantaneous maximum limits apply to results of grab samples.
2Net concentration is calculated by subtracting the influent concentration from the effluent concentration.
Table 2.NPDES permit effluent characterization study requirements for Idaho aquaculture facilities.

To comply with these new NPDES requirements in Idaho, feed ingredients must be high in protein, low in phosphorus, and very digestible. This eliminates many rendered products, such as poultry by-product meal and high-ash fish meals. High-ash fish meals, such as those produced from menhaden or seafood processing (filleting) waste, contain higher amounts of bone than fish meals made from anchovy, herring, or capelin. Bone is primarily composed of calcium phosphate, and use of highash (bone) fish meals adds too much phosphorus to feed formulations to meet the 1% total phosphorus limit.
Another approach to reducing solids production and phosphorous discharge in effluents is to increase the overall digestibility and energy content of the diet, thus decreasing the total amount of feed needed. Very high energy diets, over 30% total lipid, are being used successfully in Atlantic salmon and rainbow trout production in Europe. In Idaho, dietary lipid levels in trout feeds have increased from 12- 16% a few years ago to 20-24% today (Figure 4).
Production of fecal solids can be further reduced by pelleting feeds using extruders or expanders that cook the carbohydrate portion of the feeds, thereby eliminating the need to include indigestible binders in the feed. These feeds have improved feed conversion ratios to about 1.2:1 compared to traditional steam-pelleted feeds (1.6:1). Extruded and expanded feeds comprise an increasing portion of the aquaculture feed market in the US.
FEED INGREDIENT AVAILABILITY
The use of fish meal and fish oil in aquaculture feeds has generated controversy and become a rallying point for those who would ban certain types of aquaculture production. Total annual world fish meal production is about 6.5 million metric tons, of which about 35% is used for aquaculture feeds. While fish meal is a finite resource, average annual production has remained steady at 6.5 million metric tons over the last 10 years, even though aquaculture expanded significantly around the world.
In 1998, El Niño weather conditions caused a reduction in fish meal production of about 2 million metric tons. The increased cost of fish meal caused fish feed prices to increase. An additional factor influencing fish meal prices is China, the world’s leading aquaculture producing nation. China has become a large consumer of fish meal as it changes from polyculture to more intensive aquaculture practices that require the use of formulated feeds. Feed typically accounts for 50% of production costs. Protein sources, including fish meal, comprise 67% of ingredient costs; and if fish oil is included, over 75% of ingredient costs come from the same resource. So if another major El Niño weather pattern occurs or a shift in the market, those feeds that rely heavily on fish meal and fish oil could be subject to price increases.
Alternate protein and oil sources are an active area of nutrition research. Research focuses on in vivo measurements of nutrient availability, feed trials with alternate proteins or oils replacing fish meal and oil, and development of a rapid and accurate in vitro protein digestibility test. Alternate protein sources being investigated include wheat and corn gluten, oilseed meals and protein concentrates, rendered products, and recovered fisheries by catch and seafood processing waste. Alternate oil sources being examined include canola, flax seed, soybean, sunflower, and poultry. Alternate proteins and oils must obviously have good nutritional qualities such as high digestibility, but they must also be economically competitive, maintain product quality, and they must not increase pollutants.
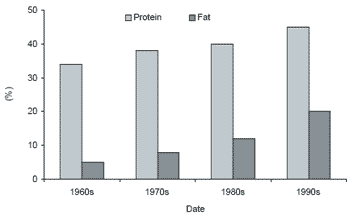
Figure 4. Changes in protein and fat levels in trout feeds from the 1960s through the 1990s (Brannon and Klontz, 1989; Fornshell, 2002).
Nutritional benefits of farmed rainbow trout
One of the advantages of aquaculture is that the nutritional and sensory quality of rainbow trout is subject to control by the farmer. Cultured rainbow trout obtain their nutrients from feed, and feed formulations can be devised to modify fillet lipid level, fatty acid profile, aroma and flavor, color, and storage stability. Fillet lipid level reflects the lipid level and the protein:energy ratio in the rainbow trout diet. Higher lipid in grow-out feeds results in higher fillet lipid levels, assuming that the feeding level remains constant. Visceral lipid stores also increase with dietary lipid content, resulting in lower yields at processing.
In rainbow trout and other fish, the fatty acid profile of storage lipids closely reflects the fatty acid profile of dietary lipids. This is an area of potential benefit to human health that is starting to be exploited by the rainbow trout industry. For example, the ω-3 fatty acid composition of trout fillets can be increased or decreased simply by altering the source of lipid added to the diet. Replacing herring oil, which is not a rich source of ω-3 fatty acids compared to many fish oils, with menhaden oil increased the percentage of ω-3 fatty acids in Atlantic salmon fillets from 20 to 27%, which is higher than in wild salmon. It must be remembered that the public health objective is to consume a target amount of ω-3 fatty acids per day. Thus the amount of ω-3 fatty acids ingested by a consumer eating a portion of rainbow trout fillet will depend both on the percentage of ω-3 fatty acids in the fillet as well as the total lipid content of the fillet. The most important point is that for farmed rainbow trout, both parameters can be readily controlled by feed composition and feeding rate during the grow-out stage.
Integrated fish health management
In rainbow trout aquaculture, host susceptibility is not just determined by immunity but is also dependent on a high quality diet and a clean water supply. It is also important to remember that these animals are cold blooded vertebrates whose immune response kinetics are determined by the temperature of their environment. While vaccines and other treatments are important for maintaining and improving fish health, so is an understanding of the role of stress, density, water quality and husbandry. The real challenge is to determine the optimum environment to maintain fish health, good growth performance and economic success. Best success has been observed when an integrated production and fish health management program is used to minimize mortality and morbidity and maintain good fish performance. Overall fish health strategies include prevention, immunization, environmental quality, nutrition, effective treatment, husbandry practices and surveillance.
As an example of a target for integrated fish health management, infectious hematopoietic necrosis virus (IHNV) is a pathogen causing significant economic loss in the commercial rainbow trout aquaculture industry in southern Idaho. Historically, infectious hematopoietic necrosis (IHN) was a chronic disease of large (100-500 g) rainbow trout in Idaho. Infected populations appeared chronically debilitated and secondary bacterial infections were common. Duration of an epizootic was 6-7 weeks with slow clinical disease that resulted in approximately 25% cumulative mortality (Busch, 1983). Currently IHN occurs in 1-10 g fish with mortality peaking in 10-14 days. Cumulative losses generally range from 1 to 50%, but may be much higher in severe cases. Although overt mortality does represent an economic impact, a more important consequence is spinal curvature (e.g. scoliosis) in 1-4% of surviving fish, making them less marketable. Additional impacts of IHN include disruption of production scheduling and increased overall production costs.
There are presently no proven or accepted methods for prevention or treatment of IHN or IHNV carriers (Winton, 1991). One measure that has been suggested, and used in some cases, to ‘control’ the spread and minimize the severity of an IHNV infection is the destruction of all infected or potentially infected fish and eggs at hatcheries. This includes fish with clinical IHN undergoing an epizootic and eggs from IHNV carrier adults.
However, this may have a greater economic impact on an aquaculture operation than the disease itself. The economic impact of a viral disease such as IHN does not just include mortality and morbidity. Reductions in food conversion and growth rate also occur. Concomitantly, water quality can deteriorate because of wasted feed, directly affecting the rearing environment and potentially the receiving water. Additionally, the full potential of selective breeding improvements cannot be realized and overall fish health maintenance is compromised.
For viral disease management, fertilized eggs are disinfected with an iodophor compound and then incubated and reared in virus-free water supplies. Iodophor disinfection of egg is sanctioned by the Food and Drug Administration as ‘low regulatory priority’. Fish up to approximately 4 g are kept in high quarantine rearing environments and prudent disinfection procedures are practiced. Whether rainbow trout are in a quarantined closed environment or in outside cement raceways, several other integrated approaches are utilized to prevent or minimize the severity of IHN. These include minimizing the impact of other pathogens, reducing handling stressors, and maintaining optimal fish densities and environmental quality.
Because there are no effective treatments or licensed vaccines for viral disease management, other strategies that have been effectively utilized by the commercial industry are bird exclusion devices. These enclosures not only prevent or minimize bird predation but they also appear to be an effective disease management tool. These ‘bird cages’ are total enclosures that prevent birds from preying upon sick fish and acting as a vector in spreading pathogens (Figure 5).
Potential new regulations
In 1989, the Natural Resources Defense Council filed a lawsuit against the Environmental Protection Agency (EPA) for lack of enforcement of the Clean Water Act. The lawsuit resulted in a settlement and Consent Decree on January 31, 1992. The Consent Decree provided for EPA to develop effluent limitation guidelines for certain specified industries and established a timetable for initiating guidelines for additional industries. EPA originally designated the Industrial Container Cleaning industry as a category for rulemaking. However, in late 1999, EPA asked the court to substitute aquaculture for the Industrial Container Cleaning industry. The reasons given by EPA for this action include the following; 1) The only relevant EPA guidance on aquaculture was over 20 years old, 2) The aquaculture industry has changed significantly in terms of the types of species raised and the industrial processes employed, and 3) Aquaculture point sources appear to discharge nutrients that states regularly identify as one of the most common causes of water quality impairment in this country. EPA has also indicated that in addition to developing effluent limitation guidelines for nutrients they would also consider effluent limitation guidelines for aquatic animal pathogens and drugs and chemicals.

Figure 5.‘Bird cages’ totally enclose rainbow trout production facilities in Idaho and prevent birds and other small animals from acting as a vector in spreading pathogens.
AQUATIC ANIMAL PATHOGENS
Huge gaps exist in our knowledge regarding pathogen distribution in the environment, the environmental fate of pathogens and host susceptibility in aquatic ecosystems. There are many misperceptions held by the public and scientific community regarding the spread of infectious agents from cultured aquatic animals to wild aquatic animals. While it is true that human activities have affected aquatic animal health through direct changes in habitat and ecosystems, these changes do not necessarily mean that the aquatic animal pathogens were, or are, actively introduced through these actions (Flagg et al., 2000). While intensive culture may magnify the effects of these pathogens within cultured populations, it does not create or establish the association of the agent and the aquatic species.
That aquaculture-related activities create and spread disease in the aquatic ecosystem is a misleading and erroneous assumption. Infectious disease agents are an integral part of existence for all animals, including both cultured and wild aquatic animal populations. In general, most of these pathogens existed in aquatic populations either prior to or in the absence of aquaculture. However, studies to examine the prevalence and the impact of infection or disease on free-ranging fish populations have been limited. Detection of infected fish and assessment of the potential impact of disease in a free-ranging population are both difficult and expensive. Issues associated with sampling free-ranging populations and the possible removal of infected fish by predators are complex. In contrast to free-ranging populations, artificial propagation of aquatic animals presents a captive population and an intensively monitored population. Captivity coupled with the routine monitoring of the health and performance of fish in aquaculture systems facilitates the identification of pathogens that have evolved with their hosts in natural environments.
Significant gaps in our knowledge of pathogen transfer are due in part to the lack of reliable, standardized or validated methods for testing effluent for aquatic animal pathogens. This is further compounded by the lack of information about pathogen amplification when an aquatic animal host, at various life stages and under different environmental conditions, becomes infected or diseased. Additionally, many characteristics of aquatic animal pathogens are poorly described yet are critical to any risk assessment. These include information on the ability of the pathogen to multiply and remain viable in water, the survival time outside the host, and the number of infectious units required to cause infection and pathogenicity.
A national Wild Fish Health Survey has been initiated by the US Fish and Wildlife Service. A survey of this type has helped to identify where certain pathogens exist and will assist in identifying pathogen geographic ranges. Additionally, this will allow comparisons among states or watersheds that may help identify why a pathogen in one area has negative effects on certain fish stocks but not on others. Most importantly, this information will provide a scientific basis for management decisions regarding stocking and fish transport activities, which has been lacking for many years. The real challenge will be to determine how to use this information in establishing new regulation and control programs or in modifying existing ones.
DRUGS AND CHEMICALS
Drugs and chemicals in aquaculture are used for therapeutic reasons, primarily in response to disease and pathogens, but are not used for growth promotion. Non-therapeutic uses are uncommonly or never used. When used, they involve selected adult individual animals in confined and controlled situations using FDA approved drugs (e.g. reproduction), or to mitigate further outbreak of disease (disinfection). Restricted and minimized use of drugs and chemicals in rainbow trout production occurs at several levels:
(a) Aquaculturists avoid the use of drugs or chemicals except where necessary for many reasons, including costs. Use of approved drugs and chemicals is the responsibility of the grower in conjunction with an aquatic animal health specialist.
(b) Quality Assurance Programs: Producers already have quality assurance programs in place that spell out the best way to maintain stocks of aquatic animals. Included in every quality assurance program is the proper and legal use of drugs and chemicals.
(c) All processors of fish and fishery products must develop and implement a FDA-required Hazard Analysis Critical Control Point (HACCP) plan. Included in all plans are requirements to ensure that there is no hazard from use of aquaculture drugs. HACCP covers use of approved drugs, drugs under investigation (INADs), extra-label use, and Low Regulatory Priority drugs by producers. The processor may get a receipt of evidence establishing that the producer operates under a third party-audited Quality Assurance Program for aquaculture drug use (US Food and Drug Administration, 1998). This type of scrutiny ensures that the producer is using only approved drugs and is not using them in a manner that will cause hazards, either for consumers or the environment. Producers take the FDA HACCP requirements very seriously and have gone to considerable effort to ensure they do not process and market any fish that have not complied with withdrawal times when drugs are used. There are no credible reports of any domestic product violating FDA standards for drug residues.
It is not known to what extent antibiotics are actually used in the US aquaculture industry. Credible estimates (MacMillan, 2001) suggest only about 31.75 mT are used in the entire US industry every year. There are only two antibiotics, oxytetracycline and Romet-30® (sulfadimethoxine plus ometoprim), licensed for use on any species of fish. This estimate has been recently corroborated by the US Geological Survey (USGS) Upper Midwest Environmental Sciences Center (UMESC, La Crosse, Wisconsin), who obtained figures on the annual use of tetracyclines from PhiBro Animal Health (current NADA sponsor of oral oxytetracycline). About 1,470 mT of tetracyclines are used in all of agriculture with about 29.5 mT used in aquaculture (i.e., ~2% of the total tetracyclines used per year). Very little Romet-30® is used in US aquaculture because of the extended 42-day withdrawal time for rainbow trout and salmon. In addition, it was discovered several years ago in the catfish industry that simply removing diseased fish was a superior treatment than feeding medicated feed.
While the estimated volume of antibiotics used is useful, there is no information on how much of that amount is actually discharged to waterways. Aquaculture systems relying on closed ponds would have essentially zero discharge; and this type of system predominates in the US (e.g. catfish industry). Flow-through aquaculture systems typically rely on sediment trapping to capture solids that might contain antibiotics. It is not known if any antibiotic in medicated feeds is solubilized. The use of antibiotics in US aquaculture has been recently reviewed (MacMillan, 2001). The role of antibiotics in US agriculture was also the subject of a recent American Academy of Microbiology colloquium report (Isaacson and Torrence, 2002). The consensus of colloquium participants was that the evaluation of antibiotic usage and its impact were complex, subject to much speculation and had a polarizing effect. EPA should not rely upon publications that report subjective and speculative statements of cause and effect in establishing effluent guidelines. It was concluded in the American Academy of Microbiology report that considerably more research would be needed to determine what role animal agriculture, including aquaculture, might have on the prevalence of human pathogenic bacteria resistance to antibiotics.
Conclusions
Market competition, water resources, feed ingredient availability and environmental constraints will limit expansion of the US rainbow trout industry. Increased market competitiveness will only occur through reducing production costs. Research in the areas of feeds and ingredients is required to enhance fish performance and identify acceptable and least cost fish meal and fish oil replacements. Tremendous gains in production efficiencies could be realized through rainbow trout strain improvement for growth and disease resistance. Biosecurity will continue to be a critical feature in any fish health management strategy. Because antibiotics will be relied upon less and less, there is a huge need for vaccines and vaccine delivery strategies along with identifying potential feed additives that could enhance innate immune defense mechanisms. And finally, advanced production and processing technologies such as automated feeding, better fish inventory control, better fish sizing techniques, and the continued development of value-added products is needed to enable rainbow trout growers in the US to increase production efficiency and intensification and effectively compete in the global marketplace.
References
Author: SCOTT E. LaPATRABrannon, E. and G. Klontz. 1989. The Idaho aquaculture industry. The Northwest Environment Journal 5:23-35.
Busch, R.A. 1983. Viral disease considerations in the commercial trout industry in Idaho. In: Workshop on viral diseases of salmonid fishes in the Columbia River Basin. (J.C. Leong and T.Y. Barila, eds). Bonneville Power Administration, Special Publication, Portland, Oregon. pgs 84-100.
Flagg, T.A., B.A. Berejikian, J.E. Colt, W.W. Dickhoff, L.W. Harrell, D.J. Maynard, C.E. Nash, M.S. Strom, R.N. Iwamoto and C.V.W. Mahnken. 2000. Ecological and behavioral impacts of artificial production strategies on the abundance of wild salmon populations. U.S. Department of Commerce, NOAA Tech. Memo. NMFSNWFSC- 41, 92 p.
Fornshell, G. 2002. Rainbow trout – challenges and solutions. Reviews in Fisheries Science 10:545- 557.
Hardy, R.W., G.C.G. Fornshell and E.L. Brannon. 2000. Rainbow trout aquaculture. In: Encyclopedia of Aquaculture. (R.R. Stickney, ed). John Wiley and Sons, New York. pgs 716-722.
Isaacson, R.E., and M.E. Torrence. 2002. The role of antibiotics in agriculture. A Report from American Academy of Microbiology. 15 p.
MacMillan, J.R. 2001. Aquaculture and antibiotic resistance: A negligible public health risk. World Aquaculture 5:49-52.
Maley, T. 1987. Exploring Idaho Geology. Mineral Land Publication, Boise, Idaho. 232 p.
National Marine Fisheries Service. 2003. http:// www.st.nmfs.gov/st1/trade/index.html. U.S. Food and Drug Administration 1998. Fish & fisheries products hazards & controls guide: Second Edition. Center for Food Safety and Applied Nutrition, Office of Seafood. Rockville, Maryland. 276 pp.
Winton, J.R. 1991. Recent advances in detection and control of infectious hematopoeitic necrosis virus in aquaculture. Annual Rev. of Fish Diseases 1:83-93.
Clear Springs Foods Inc., Research Division, Buhl, Idaho, USA




.jpg&w=3840&q=75)