Sustainable feeds for cobia aquaculture: case studies with organically certifiable protein sources
Aquafeeds have traditionally relied upon fish meals as the major provider of intact protein sources. Indeed, fish meal is often considered to represent the single most utilizable and efficient dietary protein source for carnivorous fishes. However, global production of fish meal has remained relatively static over the last 10-15 years, with supplies unlikely to increase due to severe pressure being exerted on all capture fisheries, including the stocks traditionally relied upon for fish meal and fish oil production (FAO, 2004).
The increasing lack of suitable protein sources for human consumption may result in the utilization of the fish meal stocks for the consumer market, despite the low flesh yield without the corresponding high ash component. This will further weaken future supplies of fish meal available not only for the aquafeed industry, but for the feed industry as a whole (Craig and McLean, 2005).
As aquafeeds already account for >50% of variable operating costs of intensive aquaculture operations, further constriction of supply or increases in cost due to demand will only result in progressively escalating feed costs due to the high cost associated with the protein component of aquafeeds (Bassompierre et al., 1997). For aquaculture to continue the expansion necessary to meet future global demands for seafood products, development of cost-effective and sustainable dietary formulations is mandatory (Catacutan and Pagador, 2004). The most effective and direct method of fueling this expansion is for significant reductions in the dependence of the aquafeed industry upon fish meal supplies to occur.
The aquafeed industry and researchers related to the field have been investigating alternative, less expensive, protein sources for more than 30 years. For an alternative protein source to effectively replace fish meal, certain prerequisites must be satisfied.
These protein sources must be competitively priced relative to fish meal on a unit protein basis and their inclusion cannot negatively impact fish performance in terms of production characteristics such as weight gain, animal health and welfare or final product quality. They must be sustainable and traded internationally as commodities (Hardy and Tacon, 2002).
Finally, these potential alternative proteins must not be an ecological burden, especially with respect to nitrogen and phosphorus discharge (Cho and Bureau, 1997). They must be easily handled, stored and amenable to pelleting. From an organic formulation and certification standpoint, they must not be genetically modified (Craig and McLean, 2005).
Due to the recent outbreaks of bovine spongiform encephalitis (BSE) and more recently, the H5N1 avian flu virus, many of the previously relied upon alternative protein sources from animal by-products or co-products are either no longer available or will become severely restricted from use in any animal feed in the very near future. This leaves a paucity of potential candidates as suitable alternative protein sources for the aquafeed industry. These include the pulses, oilseeds, grains, and fishery bycatch.
Soybean meal represents one of the most widely used alternative protein sources employed by aquaculture due to its global distribution, relatively low cost, good digestibility in a wide range of species, adequate amino acid profile (Figure 1) and high protein content (Storebackken et al., 2000). Indeed, investigations of soybean meal replacement in aquafeeds have been performed on over 50 species of aquacultured animals (Brown and Smith, 2002; Forster, 2002).
However, as with most alternative protein sources when replacing fish meal, the use of soybean meal in aquafeeds is not without problems, especially with higher level carnivores. Many of these species are limited in the dietary levels of soybean meal they can effectively utilize. For example, the marine carnivore cobia (Rachycentron canadum) can accept up to 40% replacement of fish meal with soybean meal with no negative impact on growth performance (Chou et al., 2004). However, at higher levels of inclusion, weight gain decreases significantly.
Other herbivorous or omnivorous species, such as tilapia and catfish, can tolerate much higher levels of soybean meal and other plant protein sources, given that their amino acid requirements, usually for methionine and lysine, are met through other sources or through dietary supplementation. Additionally, soybean and other plant-based alternative protein sources each contain a variety of anti-nutritional factors that negatively affect performance of cultured fish (Francis et al., 2001).
Naturally occurring compounds such as trypsin inhibitors, hemagglutinating agents, phytic acid, gossypol and glucosinolates are just a few of the more prominent antinutritional factors that must be accounted for in aquafeed formulations that utilize plant-based alternative protein sources if production characteristics and efficiency are not to be compromised. Relatively few detailed studies have incorporated analysis of antinutritional factors in aquafeeds containing plant-based alternative protein sources in aquacultured species (Bureau et al., 1998; El-Sayed et al., 2000; Twibell and Brown, 2000).
Single cell proteins as dietary supplements
Single cell proteins are an increasingly attractive alternative to other alternative protein sources. They include microalgae, bacteria and yeast, many of which have been incorporated into aquafeeds with varying degrees of success (Oliva-Teles and Goncalves, 2001). However, yeasts have, by far, been investigated most thoroughly as supplemental protein sources in aquafeeds (Oliva-Teles and Goncalves, 2001; Li and Gatlin, 2003; 2004; Li et al., 2005).
Single cell proteins such as yeast and yeast products are a rich source of protein, B-complex vitamins, pigments, complex carbohydrates (including glucans) and nucleotides and have been utilized mainly as supplements in animal feeds to compensate for inadequacies in nutrients in more traditional feedstuffs (Oliva-Teles and Goncalves, 2001; Olivera-Novoa et al., 2002; Cheng et al., 2004; Li and Gatlin, 2004). Other features of dietary yeasts and yeast products are their potential immunostimulating properties.
A wide variety of studies, with a broad range of species have illustrated enhanced non-specific immune activity, particularly under conditions of immunodepression and environmental stress (Lara-Flores et al., 2002; Olvera-Novoa et al., 2002; Li and Gatlin, 2004; McLean and Craig, 2004; Bagni et al., 2005; Li et al., 2005). Finally, yeast and yeast products have an additional nutrient input in the form of dietary nucleotides. While nucleic acids are formed endogenously by all animals and, thus, are not considered required nutrients (Carver, 1999), several studies have shown improvements in weight gain and feed efficiency when nucleotides are included as supplements in animal feeds (Burrells et al., 2001a; 2001b).
NuPro® as an alternative protein source
NuPro® is a yeast-based product formed from the collection of the cytoplasmic contents of the yeast cell. The resulting product is of consistent quality, relatively high protein content (>50% crude protein, dry weight basis), and balanced in terms of amino acid profile (Figure 1). Moreover, NuPro® is fully sustainable in terms of production and produces high quality pellets. Importantly, at higher inclusion levels, NuPro® acts as a binder in aquafeeds (Craig and McLean, 2005).
Previous research at the Virginia Tech Aquaculture Center (VTAC), part of the Department of Wildlife and Fisheries Sciences in Blacksburg, VA, has indicated great potential for NuPro® as an alternative protein source. Initial feeding trials with tilapia, Oreochromis nilotica, (McLean and Craig, 2004; Craig and McLean, 2005) and Pacific white shrimp (Litopenaeus vannamei) (Reid et al., 2004; Craig and McLean, 2005) showed increases in production characteristics when NuPro® was included as a protein source in aquafeeds for these species. More recent research has focused on the ability of NuPro® to replace fish meal in aquafeeds for a high level marine carnivore, the cobia (Rachycentron canadum).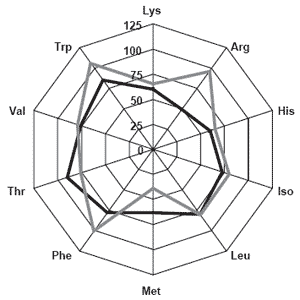
Figure 1. Essential amino acid radars of NuPro® (solid black line) and soybean meal (solid gray line) relative to Special Select® menhaden fish meal.
Cobia—a true marine carnivore Cobia is a highly prized, pelagic marine species occurring in tropical and subtropical seas except for the Eastern Pacific (Shaffer and Nakamura, 1989), and in the US are indigenous to the south Atlantic Coast and northern Gulf of Mexico (Ditty and Shaw, 1992; Franks et al., 2001). The species is an extremely popular recreational catch due to its delectable flesh and high fillet quality, and as such, demands a high market value in the seafood industry (Franks et al., 2001; Chen, 2001; Chou et al., 2001; Shiau et al., 2001), particularly in the raw fish (sashimi) market (Chou et al., 2001).
Current commercial interest in cobia culture is increasing exponentially and while certain nutrient requirements such as optimal dietary protein and lipid concentrations have been determined (Chou et al., 2001), very limited information on quantitative nutritional requirements specific for this species has been established.
COBIA RESEARCH AT THE VTAC
Nutritional research at the VTAC involving cobia has been progressing since 2002. In that time, experiments investigating protein:energy ratios, carbohydrate utilization and alternative protein utilization have been completed. More recently, the ability of cobia to utilize organically certified protein sources in replacement of fish meal has been a research priority. Lunger et al. (2006) investigated the potential of NuPro® as an alternative protein source for fish meal in aquafeeds for juvenile cobia.
In this trial, NuPro® replaced fish meal at levels of 25, 50, 75 and 100% of dietary protein (Craig and McLean, 2005). A significant impact on weight gain was observed at levels of NuPro® inclusion above 25% of dietary protein, leading to the hypothesis that cobia could potentially accept up to 40-45% inclusion of NuPro® at the expense of fish meal without detrimentally affecting production characteristics (Lunger et al., 2006). Subsequent trials repeated the 25% inclusion level of NuPro® and added a treatment where NuPro® replaced 40% of dietary protein (fish meal). An additional trial investigated whether supplementation of key essential amino acids could improve weight gain performance at higher NuPro® inclusion levels.
NUPRO® AT 25 AND 40% INCLUSION LEVELS
Juvenile cobia, initially weighing 9.9 + 0.4 g/fish were stocked in a custom designed recirculating aquaculture system, 10 fish per culture tank, for an 8 week feeding trial.
The control diet provided 45% crude protein and 10% lipid (dry weight basis), and supplied approximately 350 kcals available energy/100 g dry diet. NuPro® replaced Special Select® menhaden fish meal at 25% and 40% of dietary protein in the two experimental diets.
After 8 weeks, significant differences (P = 0.0033) were noted in weight gain and feed efficiency ratio values between fish fed the control diets and fish fed the two NuPro®-based diets (Table 1). This confirmed the results of Lunger et al. (2006) in terms of the 25% inclusion rate, and suggested that, as with soybean meal (Chou et al., 2004), cobia can accept up to 40% replacement of fish meal from alternative protein sources without detrimental impact on production characteristics.
While this is exciting from a marine carnivore standpoint, other carnivores have also been able to accept up to 40% replacement of fish meal without detrimentally affecting weight gain and feed efficiency. The increased weight gain and feed efficiency ratio values in cobia fed the NuPro® supplemented diets could have resulted due to the nucleotide content of the product, beneficially increasing the nutrient content and possibly protein absorption efficiency.
Additionally, increased levels of key essential amino acids in the 40% NuPro® diet (Figure 2) could also have resulted in superior weight gain and feed efficiency ratio values. More important is to be able to break through the 40% barrier with higher inclusion rates of alternative protein sources without affecting production. Amino acid supplementation of essential amino acids may present that very opportunity.
Table 1. Performance characteristics of juvenile cobia when NuPro® replaced fish meal at 25 and 40% of dietary protein1.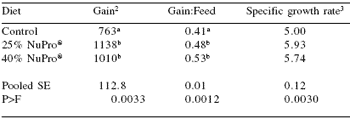
1Means of three replicate tanks.
2Percent increase from initial weight. abcMeans in the same column differ (P<0.05).
3Specific growth rate; [Ln(final wt) – Ln(initial wt.)]/42 *100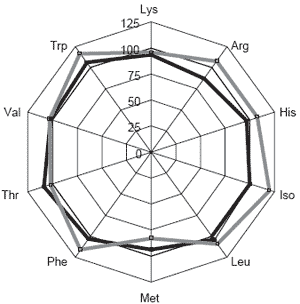
Figure 2. Essential amino acid radars for diets containing 40% NuPro® (solid black line) or 40% soybean meal (solid gray line) with 60% fish meal, relative to Special Select® menhaden fish meal at 100% dietary protein.
AMINO ACID SUPPLEMENTATION FOR TRUE CARNIVORES: TAURINE
True carnivores require taurine. Indeed, cats require taurine as one of their essential amino acids. Rainbow trout performance with feeds containing plant-based protein sources has been significantly improved when these diets were supplemented with dietary taurine (Gaylord et al., in press).
Based upon amino acid analysis of our experimental diets in the previous study with NuPro®, we found that methionine (Met) and tryptophan (Trp) could be potentially limiting based upon requirements for other marine species. Since no quantitative amino acid requirements for cobia have been determined, a feeding trial was designed to further test the upper inclusion rates for NuPro® with amino acid supplementation in aquafeeds for juvenile cobia.
In this 8 week trial, juvenile cobia initially weighing 14.5 g/fish were stocked in a custom designed recirculating aquaculture system with seven fish per 110 L aquarium. The control diet provided 44% crude protein and 12% lipid (dry weight basis), and supplied approximately 300 kcals available energy/100 g dry diet. Four experimental diets were formulated so that NuPro® replaced fish meal at 50 and 75% of dietary protein with two levels of amino acid supplementation.
In the first supplementation, Met and Trp were added (at the expense of cellulose) at levels of 0.3 and 0.2% of dry diet. The second supplementation followed the first except that taurine was added as well at a level of 0.5% of dry diet.
Diets were randomly assigned to triplicate tanks and the fish fed their respective diets twice daily (0900 and 1600 hrs) to apparent satiation. At the end of the trial, the fish fed the diets containing supplemental taurine, at both NuPro® inclusion levels, significantly (P<0.0001) outperformed those on control diets, as well as those supplemented with only Met and Trp, in terms of weight gain and feed efficiency ratio values (Table 2).
Specific growth rates were similar in cobia fed the NuPro®-supplemented diets containing taurine and the control diets (Table 2). Noteworthy from this trial are two key points: first, inclusion of NuPro® at levels higher than 40% without amino acid supplementation detrimentally affects production characteristics of juvenile cobia, again confirming the results of Lunger et al. (2006). Secondly, with amino acid supplementation, especially taurine, NuPro® inclusion level can rise to a minimum of 75% replacement of fish meal in aquafeeds for juvenile cobia without affecting production characteristics.
Although diets containing 100% fish meal replacement with NuPro® were not tested and experimental diets containing solely supplemental taurine were not investigated, these trials are currently underway. Additionally, the data certainly suggest that the amino acid deficiency resulting from NuPro® inclusion above 50% is not remedied by the supplementation of either or both Met and Trp. From these observations, it appears as if juvenile cobia may have a requirement for dietary taurine, just as do other terrestrial true carnivores.
Table 2. Performance characteristics of juvenile cobia when NuPro® replaced fish meal at 50 and 75% of dietary protein with supplemental amino acids1.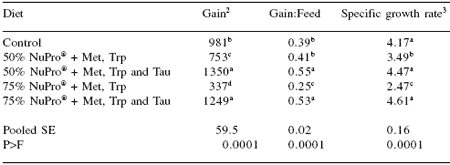
1Means of three replicate tanks.
2Percent increase from initial weight. abcMeans in the same column differ (P<0.05).
3Specific growth rate; [Ln(final wt) – Ln(initial wt.)]/42 *100
| Conclusions NuPro® certainly represents an attractive alternative protein source, not only for herbivorous and omnivorous species such as tilapia and Pacific white shrimp, but also for high level marine carnivores such as the cobia. Although amino acid supplementation is necessary at the highest inclusion levels (>50% of dietary protein), with this supplementation, especially with dietary taurine, this organically certifiable alternative protein source could provide the basis for organic production of marine carnivores, a truly novel and potentially lucrative product. Further investigation into the upper limits of NuPro® incorporation are necessary with cobia to determine whether 100% replacement of fish meal in aquafeeds designed specifically for this species is achievable. Additionally, investigations into final product quality when utilizing this alternative protein source at the highest inclusion rates are also mandatory. |
References
Authors: STEVEN R. CRAIG1,2 and EWEN MCLEAN1,3Bagni, M., N. Romano, M.G. Finoia, L. Abelli, G. Scapigliati, P.G. Tiscar, M. Sarti and G. Marino. 2005. Short- and long-term effects of a dietary yeast ß-glucan (Macroguard) and alginic acid (Ergosan) preparation on immune response in sea bass (Dicentrarchus labrax). Fish and Shellfish Immunology 18:311-325.
Bassompierre, M., A. Kjaer and E. McLean. 1997. Simulating protein digestion on trout: a rapid and inexpensive method for documenting fish meal quality and screening alternative protein sources for use in aquafeeds. Ribarstvo 55:137-145.
Brown, P.B. and K. Smith. 2002. Soybean meal in aquaculture. Soybean Meal Info Center. http://www.soymeal.org/worldlit_aquaculture.html.
Bureau, D.P., A.M. Harris and C.Y. Cho. 1998. The effects of purified alcohol extracts from soy products on feed intake and growth of chinook salmon (Oncorhynchus tshawytscha) and rainbow trout (Oncorhynchus mykiss). Aquaculture 161:27-43.
Burrells, C., P.D. Williams and P.F. Forno. 2001a. Dietary nucleotides: a novel supplement in fish feeds. 1. Effects on resistance to disease in salmonids. Aquaculture 199:159- 169.
Burrells, C., P.D. Williams, P.J. Southgate and S.L. Wadsworth. 2001b. Dietary nucleotides: a novel supplement in fish feeds. 2. Effects on vaccination, salt water transfer, growth rates and physiology of Atlantic salmon (Salmo salar L.). Aquaculture 199:171-184.
Carver, J.D. 1999. Dietary nucleotides: effects on the immune and gastrointestinal systems. Acta Paediatrics 88:83-88.
Catacutan, M.R., and G.E. Pagador. 2004. Partial replacement of fish meal by defatted soybean meal in formulated diets for the mangrove red snapper, Lutjanus argentimaculatus (Forsskal 1775). Aqua. Res. 35:299-306.
Chen, B-S. 2001. Studies on the net cage culture and disease control technology of cobia, Rachycentron canadum (Linnaeus). 6th Asian Fisheries Forum 2001: Book of Abstracts, Asian Fisheries Society. p. 41.
Chen, Y-H., M.S. Su and I.C. Liao. 2001b. Challenges and strategies of cage aquaculture development in Taiwan. Aquaculture 2001: Book of Abstracts, World Aquaculture Society. p. 373.
Cheng, Z.J., R.W. Hardy and N.J. Huige. 2004. Apparent digestibility coefficients of nutrients in brewer’s and rendered animal by-products for rainbow trout (Oncorhynchus mykiss (Walbaum)). Aqua. Res. 35:1-9.
Cho, C.Y. and D.P. Bureau. 1997. Reduction of waste output from salmonid aquaculture through feeds and feeding. Progressive Fish Culturist. 59:155-160.
Chou, R.L., B.Y. Her, M.S. Su, G. Hwang, Y.H. Wu and H.Y. Chen. 2004. Substituting fish meal with soybean meal in diets of juvenile cobia, Rachycentron canadum. Aquaculture 229:325-333.
Chou, R-L., M.S. Su and H.Y. Chen. 2001. Optimal dietary protein and lipid levels for juvenile cobia (Rachycentron canadum). Aquaculture 193:81-89.
Craig, S.R. and E. McLean. 2005. The organic movement: a role for NuPro® as an alterative protein source. In: Nutritional Biotechnology in the Food and Feed Industry (KA Jacques and TP Lyons, eds). Nottingham University Press, UK.
Ditty, J.G. and R.F. Shaw. 1992. Larval development, distribution, and ecology of cobia Rachycentron canadum (family: Rachycentridae) in the northern Gulf of Mexico. Fishery Bulletin. 90:668-677.
El-Sayed, A-F.M., I. Martinez and F.J. Moyano. 2000. Assessment of the effect of plant inhibitors on digestive proteases of Nile tilapia using in vitro assays. Aquaculture International 8:403-415.
FAO. 2004. State of World Fisheries and Aquaculture 2004. FAO, Rome, Italy. Forster, I. 2002. Use of soybean meal in the diets of non-salmonid marine fish. United Soybean Board. American Soybean Association. http://www.soyaqua.org/.
Francis, G., H.P.S. Makkar and K. Becker. 2001. Antinutritional factors present in plant-derived alternate fish feed ingredients and their effects in fish. Aquaculture 199:197-227.
Franks, J.S., J.T. Ogle, J.M. Lotz, L.C. Nicholson, D.N. Barnes and K.M. Larsen. 2001. Spontaneous spawning of cobia Rachycentron canadum, induced by human chorionic gonadotropin (HCG), with comments on fertilization, hatching and larval development. Proceedings of the Gulf and Caribbean Fisheries Institute. 52:598-609.
Gaylord, T.G, A.M. Teague, and F.T. Barrows. (in press). Taurine supplementation of all-plant protein diets for rainbow trout (Oncorhynchus mykiss). Journal of the World Aquaculture Society.
Hardy, R.W. and A.G.J. Tacon. 2002. Fish meal: historical uses, production trends and future outlook for sustainable supplies. In: Responsible Marine Aquaculture. (R.R. Stickney and J.P. McVey, eds.) CAB International, Oxon, UK, pp. 311-325.
Lara-Flores, M., M.A.Olvera-Novoa, B.E. Guzmán-Méndez and W. López-Madrid. 2002. Use of the bacteria Streptococcus faecium and Lactobacillus acidophilus, and the yeast Saccharomyces cerevisiae as growth promoters in Nile tilapia (Oremochromis niloticus). Aquaculture 216:193-201.
Li, P., G.S. Burr, J. Goff, K.W. Whiteman, K.B. Davis, R.R. Vega, W.H. Neill and D.M. Gatlin III. 2005. A preliminary study on the effects of dietary supplementation of brewers yeast and nucleotides, singularly or in combination, on juvenile red drum, Sciaenops ocellatus. Aquacult. Res. 36:1120-1127.
Li, P. and D.M. Gatlin III. 2003. Evaluation of brewers yeast (Saccharomyces cerevisiae) as a feed supplement for hybrid striped bass (Morone chrysops x M. saxatilis. Aquaculture 219:681-692.
Li, P. and D.M. Gatlin III. 2004. Dietary brewers yeast and the prebiotic Grobiotic™ AE influence growth performance, immune responses and resistance of hybrid striped bass (Morone chrysops x M. saxatilis) to Streptococcus iniae infection. Aquaculture 2319:445-456.
Lunger, A., E. McLean and S.R. Craig. 2006. Replacement of fish meal in cobia (Rachycentron canadum) diets using an organically certified protein. Aquaculture (in press).
McLean, E. and S.R. Craig. 2004. Growth performance of Nile tilapia fed an organically certified yeast-based alternative protein source. In: Proceedings of the 5th International Conference on Recirculating Aquaculture. (G.J. Flick, A. Correa, T. Rakestraw, eds.) July 24-27th 2004, Roanoke, Virginia, USA, pp. 580-586.
Olivera-Novoa, M.A., C.A. Martinez-Palacios and L. Olivera-Castillo. 2002. Utilization of torula yeast (Candida utilis) as a protein source in diets for tilapia (Oreochromis mossambicus Peters) fry. Aquaculture Nutrition 8:257-264.
Oliva-Teles, A. and P. Goncalves. 2001. Partial replacement of fish meal by brewers yeast (Saccaromyces cerevisiae) in diets for sea bass (Dicentrarchus labrax) juveniles. Aquaculture 202:269-278.
Reid, B., E. McLean and S.R. Craig. 2004. Performance characteristics of shrimp (Litopenaeus vannamei) fed a certified organic feed versus an investigational organic aquafeed. In: Proceedings of the 5th International Conference on Recirculating Aquaculture (G.J. Flick, A. Correa, T. Rakestraw, eds.) July 24-27th 2004, Roanoke, Virginia, USA, pp. 539-542.
Shaffer, R.V. and E.L. Nakamura. 1989. Synopsis of biological data on the cobia, Rachycentron canadum, (Pisces: Rachycentridae). U.S. Department of Commerce, NOAA Tech. Rep. NMFS 82 (FAO Fish Synop. 153). 21 pp.
Shiau, C-Y., R-T. Lin, Y-L. Li and T-K. Tze. 2001. Characterization of chemical composition of cage cultured cobia Rachycentron canadum. 6th Asian Fisheries Forum 2001: Book of Abstracts, Asian Fisheries Society. p. 338.
Storebakken, T., S. Refstie and B. Ruyter. 2000. Soy products and fat and protein sources in fish feeds for intensive aquaculture. In: Soy in Animal Nutrition. (J.K. Drackley, ed.), Fed. Animal Sci. Soc., Savoy, IL. pp. 127-170.
Twibell, R.G. and P.B. Brown. 2000. Utilization of soy protein in fish feeds. Feed Tech. 4:43-45.
1 Virginia Tech Aquaculture Center, Blacksburg, Virginia;
2 Department of Large Animal Clinical Sciences, Virginia Polytechnic Institute and State University, Virginia-Maryland Regional College of Veterinary Medicine, Blacksburg, Virginia;
3 Department of Fisheries and Wildlife Sciences, Virginia Polytechnic Institute and State University, Blacksburg, Virginia



.jpg&w=3840&q=75)