Water quality is determined by some variables like temperature, transparency, turbidity, water colour, carbon dioxide, pH, alkalinity, hardness, unionized ammonia (NH3), nitrite and nitrate (Saeed, 2013; Saeed and Abdel-Mageed, 2011; Chandra Das et al., 2005). For improving water quality and decreasing ammonia toxicity, there are several possible methods including zeolite addition (U.S.EPA, 2007; Shalaby, 2007). Zeolite addition has been used successfully in freshwater systems to decrease ammonia concentrations and toxicity for several decades. However, zeolite in marine systems has been used less because ions in the seawater interfere with zeolite’s ability to adsorb ammonia (Burgess et al., 2004). Constantly ongoing research shows promising opportunities for using natural and modified zeolites to purify wastewaters and environment protection, because of their unique molecular sieve, sorption and ion exchange properties (Kedziora et al., 2014).
Natural zeolites represent a large and very diverse group of minerals such as water-silicates that are characterized by three-dimensional structure and belong to the class of aluminum silicates from a chemical point of view (Strakova et al., 2008; Vaughan, 1988). Additionally, natural zeolites are porous and contain hydrated aluminum silicates that have ion-exchange and adsorption properties and have a large surface area that helps in the adsorption properties (Xia et al., 2009). Moreover, zeolite can be used as an antimicrobial agent (Haile and Nakhla, 2010).
Forms of zeolite have been used to remove ammonium from aqueous solutions (Wen et al., 2010). Zeolites are used in industry, agriculture, environment protection and even in medicine. Zeolites have a relatively high Si/Al compositional ratio which gives it the special ion-ex-change selectivity for large monovalent cations. Natural or synthetic zeolites (sodium aluminum silicates) are known to ease adsorb metal ions by exchange reactions (Jain, 1999). So, the natural zeolites, clinoptilolite may be useful as metal scavengers in metal-rich sludges to its ability to take up heavy metals; Cd, Cr, Cu, Fe, Mn, Ni, Pb, Zn (Zorpas et al., 2000).
As good water quality of fish culture ponds and aquatic environment play a critical role for successful fish production, water quality management in fish ponds is a determent step (Saeed, 2013). Thus, natural zeolites can apply successfully in aquaculture to remove ammonium from hatchery, transport, aquarium waters, generate oxygen for aeration systems in aquaria, transport and additives fish feed. Zeolite cation exchange removes NH4 + from recirculating fish production systems and hatcheries waters produced by the decomposition of excrement and/or unused food, much as NH4 + is removed from municipal sewage effluent and wastewaters (Bruin et al., 1980; Piper and Smith, 1984). Therefore, the present study was conducted to evaluate the impact of adding natural zeolite for improving water quality of fish culture, growth performance and health status of Nile tilapia (Oreochromis niloticus) fish in earthen ponds.
MATERIALS AND METHODS
Experimental Design.
The study had been done in a private farm at Tollumbat No. 7 (agriculture drainage water) in Riyad City, Kafr El-Sheikh governorate, Egypt. Water, sediment and fish samples were collected from 6 shallow (1feddan each and 1.25 m deep) earthen ponds represent three treatments (two ponds each). The first treatment (control) was under normal fish culture conditions without zeolite addition, and the second treatment (T1) in which natural zeolite was scattered on the pond bottom (about 500kg\feddan), while in the third treatment (T2) natural zeolite was used as a mechanical filter where zeolite (500kg) was placed in a concrete pond with dimensions (1.5m length, 2.30m width and 90cm height) in which the irrigation water passed through it and then distributed to the ponds by water inlet pipes. Natural zeolite as shown in Figure 1 are porous granules of sedimentary rocks which have the ability to gain and lose water with great ability for ions exchange.
The ponds were stocked with monosex males of Nile tilapia, Oreochromis niloticus fingerlings at an average initial length of about 11.40, 11.50 and 12.10 cm and an average initial weight of 22.90, 23.50 and 25.70 g for control, T1 and T2, respectively. Each pond was stocked with 12000 fish/feddan (3 fish/m2). Nile tilapia fingerlings were fed on a commercial diet containing 25% crude protein six days/week at a daily feeding rate of 3% of an average fish-body weight twice at 9.0 am and 3.0pm during the experimental period in each pond. The trial lasted for 22 weeks and at the end of the experiment, ponds were drained and fish were harvested, counted, and the individual weight and length were measured and also water and sediment were analyzed.
Figure 1. Commercial natural zeolite (Clinoptilolite) granules.
Sampling:
Water, sediment were collected monthly and fish samples biweekly from each pond. Water samples were taken from different places at each site by a PVC tube column sampler at depth of half meter from the water surface. The samples at each site were mixed in a plastic bucket and a sample of 1 liter was placed in a polyethylene bottle, kept refrigerated and transferred cold to the laboratory for analysis. Sampling of bottom sediments (from the upper 10 cm surface layer) using Peterson grab as described by Boyd and Tucker (1992) and kept in cleaned plastic bags. The wet sediment samples were air dried, pulverized, and then mixed to provide a homogeneous mixture. Three sediment replicate samples from each site were combined. Nile tilapia freshwater fish were collected and transported to the laboratory in an ice box, where tissue samples were taken.
Laboratory Analysis.
a. Water:
Hydrogen ion concentration (pH) was measured with pH meter (Model 25, Fisher Scientific). Total dissolved solids (TDS as g/l) were determined using a salinity-conductivity meter (model, YSI EC 300). Temperature and dissolved oxygen were measured by using a digital oxygen meter (Model YSI 55). The concentration of total hardness (mg/l as CaCO3), total ammonia (NH4-N+NH3- N), unionized ammonia (NH3-N), nitrite (NO2-N) and nitrate (NO3-N) were measured by methods described in Boyd and Tucker (1992). Transparency (cm) was measured by using a Secchi Disc of 20cm diameter. Heavy metals were extracted with conc. HNO3 and HCl and preserved in a refrigerator till analysis for Fe, Zn, Mn, Cu, Cd and Pb concentration as μg/l (EPA. 1992).
b. Metals residues in fish and sediments:
Fish, musculature and organs (gills and liver) were collected separately and metals were extracted by HNO3 and HCl according to the methods described in AOAC (1990). Sediment samples were extracted with HNO3, HCl acid and H2O2 according to EPA (1996). Atomic Absorption Spectrophotometer (Model Thermo Electron Corporation) instrument was used to detect metals concentrations which were expressed as μg/g dry wt. in both sediments and fish samples.
The degree of metal removal (%) (Removal efficiency or Purification rate %) from the investigated treatments was calculated by the following general mathematical equation;
Where CI and CT are concentrations (mg/L) in control and zeolite treatments, respectively.
c. Growth parameters:
Random samples (30 fish from each pond-60 fish for each treatment) were taken biweekly during the experimental period. Body measurements (body weight in g and body length in cm) were conducted at biweekly intervals throughout the whole experimental period growth parameters were calculated as follows:
i. Daily weight gain (DWG) was calculated using the formula:
DWG = [Average W2 (g) – Average W1 (g)] / t
Where W1 and W2 = the initial and final fish weight, while t is the experimental period in days.
ii. Specific growth rate (SGR) was calculated according to Jauncey and Rose (1982) as:
SGR = (Ln W2 – Ln W1 x 100) / t
Where, w1 = first fish weight in grams, w2 = final fish weight in grams, t = period in day.
iii. Fish condition factor (K) was calculated according to Schreck and Moyle (1990) as:
K = (Wt/L3) x 100
Where, Wt is the total gutted weight of the fish (g), and L is the total length (cm)
d. Chemical composition:
At the end of the experiment, six fish from each pond exposed to the chemical composition (moisture, protein, fat and ash content) of the whole fish body according to AOAC (2000).
e. Clinical examinations:
Clinical examinations of infested or infected fish were done according to Stoskopf (1993); for skin darkening, discoloration or paleness, skin congestion, ragged or torn fins, raised scales, haemorrhage and erosions. The abdomen was examined for enlargement and or distention.
Statistical Analysis.
One-way ANOVA and Duncan’s multiple range tests were used to evaluate the significant difference of the data which were computed by applying the computer program (SAS, 1996). Significant differences are stated at P<0.05. Data were statistically analyzed according to Bailey (1981).
RESULTS AND DISCUSSION
Physico-Chemical Parameters.
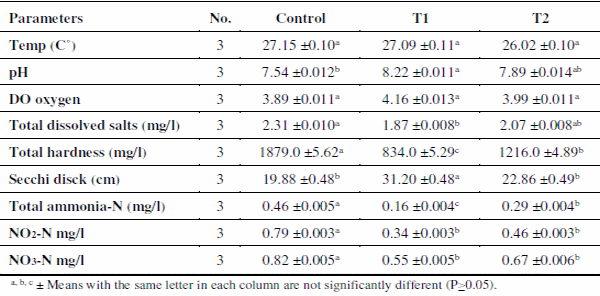
Water quality parameters are presented in Table 1. Water temperature not significantly varied among treatments throughout the experimental period and ranged from 26.02 Co in T2 to 27.15 Co in control. Application of zeolite caused a significant increase (P<0.05) of water pH (8.22 in T1 and 7.89 in T2) compared with control ponds (7.54). This may be related to that zeolites, in general, are weakly acidic in nature and sodium-form exchangers are selective for hydrogen, which leads to high pH values when the exchanger is equilibrated with relatively dilute electrolyte solutions (Leinonen and Lehto, 2001). Both carbonate and nitrate ions are attracted by the negative charge within zeolites (Mumpton, 1999). The desirable pH range for most fish species is 7-9 (Boyd, 1998).
Dissolved oxygen (DO) was not affected significantly by treatments (P>0.05) and it was higher (4.16 mg/l) in T1 followed by T2 (3.99 mg/l) than that of control (3.89 mg/l). DO attained lower value in control. The higher values of DO in T1 and T2 may be attributed to good water quality conditions caused by zeolite addition, where uneaten food and nutrients were decreased. Ferdous et al. (2013) found that DO content increased significantly after zeolite treatment. However, DO was still lower than that recommended by Boyd (1998), who advised 5.0 mg/l to satisfies the needs of fish welfare.
Regarding, total dissolved salts (TDS), it was obviously higher in control ponds (2.31 g/l) than those in zeolite treated ponds T1 (1.87 g/l) and T2 (2.07 g/l). There was a significant (P<0.05) variations in water TDS among treatments. The reduction in TDS values in T1 and T2 ponds revealed that zeolite highly absorbed various salts from pond water (Mumpton, 1999). Water hardness increased in control with a total mean of (1879.0 mg/l). On the other hand, it was decreased in T1 (834.0 mg/l) and T2 (1216.0 mg/l). This possibly could be from the adsorption activity of zeolite for removing divalent cations (Ca++, Mg++ and/or Fe++) from water (Mumpton, 1999). In addition to iron removal, zeolites can be used for water softening, through Ca2+ ion removal and replaced by ions of Na+ (Nazarenko and Zarubina, 2013).
Table 1. Some water quality parameters in different treatments during the experimental period based on means±SE.
Secchi disc readings were positively affected by treatments (P<0.05). The average values of transparency were generally higher in T1 (31.20 cm), whereas T2 showed Secchi disc readings (22.86 cm) compared with lower secchi disc reading of control ponds (19.88 cm). This resulted from the purification role of zeolite which was consistent with the results of Ferdous et al. (2013) who observed that zeolite increase water transparency. Application of zeolite significantly decreased all the inorganic dissolved nitrogen, namely total ammonia (NH3-N+ NH4 +-N), nitrite (NO2-N) and nitrate (NO3-N) content in the water. The levels of these nutrients in the three treatments were significantly (P<0.05) different with higher values in control ponds (0.46, 0.79 and 0.82 mg/l, respectively).
Significantly lower concentrations of nitrogen nutrients in T1 (0.16, 0.34 and 0.55 mg/l, respectively) and T2 (0.29, 0.46 and 0.67 mg/l, respectively) might have been principally due to the adsorption capacity of zeolite as it attracted nitrate ions and improve nutrient retention from soil and water (Mumpton, 1999). Also, Polat et al. (2004) mentioned that zeolite clinoptilolite retains valuable nitrogen. The negative charge on zeolite allows for the adsorption of certain positively charged ions as NH4 + which is preferentially adsorbed to the zeolite matrix. Durhan et al. (1993) and Besser et al. (1998) mentioned that zeolite has been used to decrease the concentrations of ammonium in freshwater. A direct relationship between the mass of zeolite and total ammonia removal was evident (Burgess et al., 2004). The desirable ranges are 0.2-2.0 mg/l for total NH4-N, <0.3 mg/l for NO2-N and 0.2-10 mg/l for NO3- N in fish ponds (Boyd, 1998).
Heavy Metals.
a. Water:
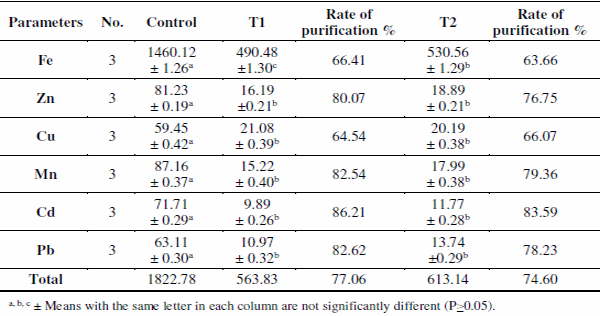
Metals concentration values showed significant differences (P<0.05) for the three treatments (Table 2), in which, control ponds had the highest total mean concentrations of all metals (1822.78 μg/l) compared to T1 (563.83 μg/l) and T2 (613.14 μg/l). Hamed et al. (2013) attributed the increase of metals content in water to the decomposition of organic matter in sediments and release of metals to the overlying water. Also, Fe showed the highest accumulated metal in different sites, while Pb was the lowest one.
Natural zeolite, clinoptilolite has the ability to take up heavy metals; Cd, Cu, Fe, Mn, Pb and Zn (Barloková and Ilavsky, 2010). Thus, application of zeolite (clinoptilolite) decreased heavy metals in pond's water and was more effective in T1 which comply with studies of showed that natural zeolites were able to remove cationic heavy metal elements from industrial wastewater. Moreover, Kocakusak et al. (2001) mentioned that zeolites application in wastes increased both heavy metals and ammonium ions removal. Metal ions’ removal efficiencies for tested pond's water treated with zeolite are shown in Table 2. The degree of metal removal (%) (Removal efficiency of ions or Purification rate %) from the investigated treatments using natural zeolite was calculated by the following mathematical formula;
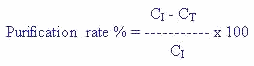
In which, zeolites had selectivity chelated Cd, Pb, Mn and Zn. In the two treatments the selectivity of ions was: Cd> Pb> Mn> Zn> Fe> Cu. The difference in selectivity might be attributed to ion-exchange processes. Also, data demonstrated the preference of zeolite for Cd and Pb compared to other metals. Similar results were recorded by Shaheen et al. (2012) and Mieret et al. (2001) who describing more interactions of Pb2+ and Cd2+ competing for ionexchange sites in natural zeolite; clinoptilolite.
Table 2. Means±SE of heavy metals concentration (μg/l) in water of different treatments.
Zeolite, clinoptilolite as a bulking material has the ability to increase the porosity of the substrate and as a result, to improve the composting process and the biodegradability of the organic matter (Wagner et al., 1990). The net negative charge in zeolite is balanced by the positive cations (sodium, potassium, or calcium). These cations are exchangeable with certain cations in solutions such as lead, cadmium, zinc, and manganese (Barrer, 1978) and make zeolite suitable for removing undesirable heavy metal ions from industrial effluent waters.
b. Sediments:
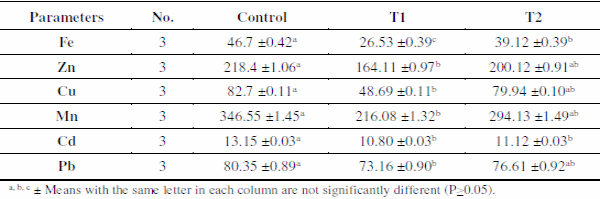
Results of heavy metals in sediments are presented in Table 3. The mean concentrations of metals differed between sediments of the three treatments and were detected in the following order: Fe> Mn> Zn> Pb> Cu> Cd. These results showed that sediments of control ponds accumulate high levels of all studied metals. The distributions of metals are associated mainly with the organic matter fraction of sediments as found by Saeed and El-Gammal (2009), who also observed a significant positive correlation between organic matter and each of Fe, Zn, Cu and Mn and no correlation with Pb.
The increases of Fe and Mn could be attributed to their increase in the earth's crust. Zinc and Cu accumulation could come from feed as recorded by Wudtisin and Boyd (2006). They also suggested that the accumulation of Fe, Mn, Cu, and Zn could have occurred by precipitation with hydroxyl ions and carbonates under aerobic conditions. The pH increase causes precipitation of hydrous ferric oxides and decreased dissolved metal concentrations as metals precipitate as hydroxides (Wingenfelder et al., 2005). The ability to exchange cations and adsorb inorganic and organic molecules of certain sizes and catalytic properties are among the most significant features of zeolites. So, using the natural material zeolite is one of the methods to make iron and manganese removal more effective (Barloková, 2008).
Table 3. Means±SE of heavy metals concentrations (μg/g dw except Fe mg/g dw) in sediments of different treatments.
c. Fish:
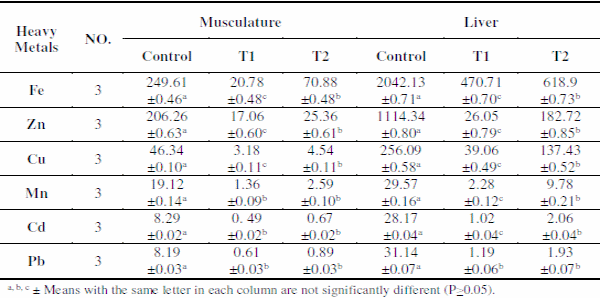
Metals accumulation in musculature and liver of O. niloticus in different treatments are shown in Table 4. All metals showed significant (P<0.05) difference among different treatments. It was observed that the concentrations of all studied metals in fish reared in T1 and T2 were lower than those reared in control ponds. This may be explained by the assumption that dissolved metals are adsorbed on zeolite (Shaheen et al., 2012), thus leads to decreasing metals bioaccumulation in fish.
Table 4. Means±SE of heavy metals concentration (μg/g dry wt.) in musculature and liver of O. niloticus in different treatments.
The abundance of heavy metals in different tissue/organs showed that liver had a high tendency to accumulate heavy metals than musculature. This may be explained as liver is the main target organ for detoxification and excretion of toxicants besides gills which being the key interface for the uptake of waterborne metal from water (Noegrohati, 2006).
Growth Parameters.
Growth in the fish can be simply monitored by measuring the increase of fish weight and length traits. Condition factor as an index of growth provides a measure of fatness "plumpness" or "robustness" of fish and food conversion efficiency. Condition factor is frequently assumed to reflect not only characteristics of fish such as health, reproductive state and growth, but also characteristics of the environment such as water quality (Schreck and Moyle, 1990).
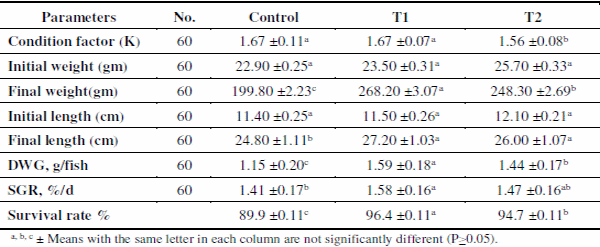
Concerning the values of condition factor (k) of O. niloticus (Table 5) for different treatments; were 1.67, 1.67 and 1.56, respectively. Control ponds and T1 had the highest (P<0.01) K value compared to T2. Fish reared in T1 showed the highest (P<0.01) final body weight followed by T2 then control ponds (268.20, 248.30, and 199.80g), respectively. Average fish final length at the end of the experimental period was found to follow the same order (P<0.01) of final body weight and being 27.2, 26.0 and 24.8 cm, respectively.
Regarding the survival rate of Nile tilapia, it improved (P<0.05) with both types of application addition either in the pond bottom or used as a filter. The highest value of survival (96.4%) was obtained with addition of zeolite in the pond bottom (T1), followed by filter zeolite (T2), while the lowest (89.9%) was observed for control. These results are in accordance with those obtained by Osman et al. (2008), who found a strong correlation between body weight and body length for tilapia. The fluctuations in fish growth (length and weight) are affected by different factors such as feeding regime (Saeed and Abdel-Mageed, 2011) and environmental conditions (Saeed, 2013).
On the other hand, the highest averages daily gain and SGR were recorded at T1 (1.59 and 1.58%/day, respectively) followed by T2 (1.44 and 1.47%/day, respectively), while the lowest values (1.15 and 1.41%/day, respectively) were found in control ponds. The good conditions of water quality in T1 and T2 (treated with zeolite) may be a reason for higher harvest weights than those reared in control ponds. The data in the present study matches the conclusions of Xia et al. (2009) who reported that natural zeolite helps to prevent the occurrence of disease and enables to promote growth and survivability and higher profits.
Chemical Composition of Fish.
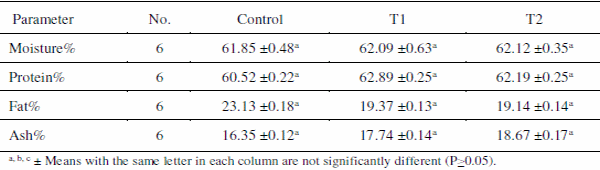
The results of proximate analysis of the whole fish body of O. niloticus for moisture, protein, fat and ash are shown in Table 6. Its averages were 61.85 % moisture, 60.52 % protein, 23.13 % lipid and 16.35 % ash in control ponds. The corresponding values of fish in T1 were 62.09% moisture, 62.89 % protein, 19.37 % lipid and 17.74 % ash. The values for fish in T2 were 62.12% moisture, 62.19 % protein, 19.14 % lipid and 18.67 % ash content. It is clear that application of zeolite had no significant effect on the proximate composition of the fish. This may be that all fish in all ponds fed artificial feed. According to Hanley (1991), endogenous factors (size, sex and stage of life cycle) and exogenous factors (diet composition, feeding frequency, temperature etc.) affect the body composition of fish.
Table 5. Means±SE of some growth parameters of O. niloticus in different treatments.
Table 6. Proximate chemical analyses (dry wt. basis) of O. niloticus reared in different treatments.
Clinical Examinations of Fish.
The clinical examination of some moribund and mortalities from all groups showed that some aggregated on the surface, accumulated at the water inlet and appeared dull, lethargic with loss of escape reflex. On the other side, the control group showed yellowish discoloration of the skin, dermatitis surrounded with petechial hemorrhage at the peduncle region indicating liver failure (jaundice like appearance) and less microbial resistance. This may be due to the pollution of the source of water supplying fish farms with some industrial wastes reaching the agriculture drainage wastewater used for aquaculture in Egypt (Mohana, 1996; Fajardo, 2002). The improved production of treated groups could be as a result of the ability of zeolite to adsorb heavy metals and other pollutants like ammonia besides its antimicrobial effect (Haile and Nakhla, 2010).

Figure 2. Some fish of the control group suffering yellowish discoloration, dermatitis surrounded with petechial hemorrhages at the peduncle region and at the fin base.
In conclusion, application of zeolite in fish ponds improved fish health, disease resistance, water quality parameters and decreased heavy metals accumulation in water, sediment and in organs as well as improved growth parameters. However, proximate chemical composition of fish musculature did not significantly vary. Application of zeolite scattered on the pond bottom increased the efficiency of improving pond quality than used as a filter.
This article was originally published in Abbassa Int. J. Aqua., V0l.8 No.(2) 2015. Pags. 361-383.

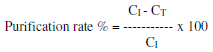
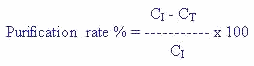








.jpg&w=3840&q=75)