
Content sponsored by:
Hangzhou DE Mark Industrial Co Ltd
Vitamin Highlights
Published: September 11, 2018
Source : Ronnick FONG. Huzhou International Trade Co. Ltd.
Vitamin B6
Guangji Pharmaceutical Holding Co., Ltd. announced on August 16 the resumption of production of vitamin B6.
On September 18, 2017, Guangji Pharmaceutical’s subsidiary Hubei Huisheng Pharmaceutical Co., Ltd. (Huisheng Pharma) shut down production due to fire in the workshop. The resumption works were started on March 22, 2018, and completed at the end of June. The relevant production process has been optimized and upgraded. After trial production, it is proved that the modified production line meets the production needs. However, taking into account the high temperatures in July/ August, Huisheng Pharmaceutical decided to temporarily postpone production in response to market conditions and other factors.
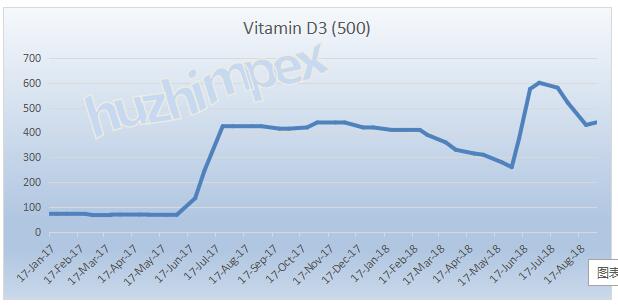
Vitamin D3
Some manufacturers in June have significantly increased their quotations, but the demand is not satisfying, and the recent market has oscillated and the price went back. On August 24, Taizhou Haisheng Company increased the price of feed grade vitamin D3 products to 1,000 yuan / KG..
Antibiotics Update
Ningxia Tairui Pharma Punished for Unauthorized Production
Ningxia Tairui Pharma is severely punished as long as the production is resumed without authorization.
On June 22, 2018, according to the local government's notice of suspension of production, the producing workshops of Ningxia Tairui Pharmaceutical Co., Ltd. (Tai Rui Pharma) was fully discontinued and rectified. At the same time, the Company announced and promised to fully implement the relocation plan. In mid-July, Tairui Pharma commissioned the fermenter to verify the effect of odor management. In response to the complaints from nearby residents, the Environmental Protection Inspectors promptly requested Tairui Pharma to suspend production and will further verify the company's situation. As of August 18, all seven fermenters that were resumed production were completely emptied and sealed.
Extended and updated:
1---According to the incident, the heads of both city and county level were dismissed and the other responsible officials were seriously accountable. The chairman of Tairui Pharma has been detained for alleged environmental crimes.
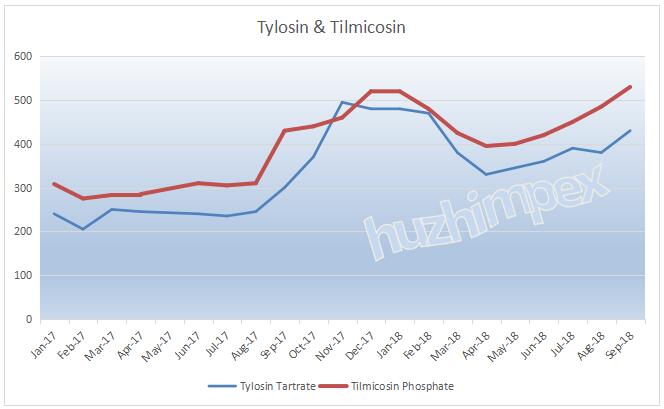
2---The global tylosin manufacturers are mainly Ningxia Tairui Pharmaceutical, Lukang Medicine and Qilu Pharmaceutical. Ningxia Tairui Pharma is currently the world's largest manufacturer of tylosin, with a production capacity of over 5,000 tons, and its market share of tylosin and tilmicosin is 63% and 54% respectively. The suspension of Ningxia Tairui Pharma and the limited production of Qilu Pharmaceutical have resulted in continued supply tension of tylosin, which has a seriously negative impact to the market. The prices of tylosin, tilmicosin and tiamulin will continue to go upward at high level.
Source
Ronnick FONG. Huzhou International Trade Co. Ltd.Related topics
Join to be able to comment.
Once you join Engormix, you will be able to participate in all content and forums.
* Required information
Would you like to discuss another topic? Create a new post to engage with experts in the community.
Create a post